UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM
(Mark One)
For
the fiscal year ended:
For the transition period from [ ] to [ ]
Commission
file number
(Exact name of registrant as specified in its charter)
(State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification No.) |
(Address of principal executive offices)
Registrant’s
telephone number: (
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) |
Name of each exchange on which registered | ||
| The Stock Market LLC (The Nasdaq Capital Market) | ||||
| The
|
Securities registered pursuant to Section 12(g) of the Act:
None
Indicate
by check mark if registrant is a well-known seasoned issuer, as defined under Rule 405 of the Securities Act. Yes ☐
Indicate
by check mark if registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐
Indicate
by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange
Act during the preceding 12 months (or for such shorter period that the issuer was required to file such reports), and (2) has been subject
to such filing requirements for the past 90 days.
Indicate
by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule
405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant
was required to submit such files).
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer | ☐ | Accelerated filer | ☐ |
| ☒ | Smaller reporting company | ||
| Emerging growth company |
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act.
Indicate
by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness
of its internal control over financial reporting under section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered
public accounting firm that prepared or issued its audit report.
If
securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant
included in the filing reflect the correction of an error to previously issued financial statements.
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate
by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Act). Yes ☐ No
As
of June 30, 2025, the aggregate market value of the common stock of the registrant held by non-affiliates was approximately $
The number of shares of the registrant’s common stock outstanding as of March 18, 2026 was .
Documents incorporated by reference
TABLE OF CONTENTS
| 2 |
In this report, unless the context indicates otherwise, the terms “Company,” “we,” “us,” “our” and similar words refer to BullFrog AI Holdings, Inc. (“BullFrog”), a Nevada corporation.
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This report contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, or the “Securities Act,” and Section 21E of the Securities Exchange Act of 1934 or the “Exchange Act.” These forward-looking statements are subject to certain risks and uncertainties that could cause actual results to differ materially from historical results or anticipated results.
In some cases, you can identify forward-looking statements by terms such as “may,” “intend,” “might,” “will,” “should,” “could,” “would,” “expect,” “believe,” “anticipate,” “estimate,” “predict,” “potential,” or the negative of these terms. These terms and similar expressions are intended to identify forward-looking statements. The forward-looking statements in this report are largely based upon management’s current expectations and beliefs about future events and trends affecting our business, which management believes are reasonable. These forward-looking statements are subject to risks and uncertainties that could cause actual results to differ materially from those anticipated in the forward-looking statements. In addition, we cannot assess the impact of each factor on our business or the extent to which any factor or combination of factors, or factors we are unaware of, may cause actual results to differ materially from those contained in any forward-looking statements. You are cautioned not to place undue reliance on any forward-looking statements. These statements represent our estimates and assumptions only as of the date of this report. Except to the extent required by federal securities laws, we undertake no obligation to update any forward-looking statement to reflect events or circumstances after the date hereof or to reflect the occurrence of unanticipated events.
You should be aware that our actual results could differ materially from those contained in the forward-looking statements due to a number of factors, including:
| ● | our future financial performance, including our revenue, costs of revenue, operating expenses and profitability; |
| ● | the sufficiency of our cash and cash equivalents to meet our liquidity needs; |
| ● | our predictions about, and the development of, digital transformation technology and bio health businesses and their respective market trends; |
| ● | our ability to attract and retain customers for our products and services; |
| ● | the availability of financing for smaller publicly traded companies like us; |
| ● | our current and future capital requirements to support the continued development and commercialization of our products and services; | |
| ● | our ability to maintain compliance with Nasdaq listing rules; |
| ● | our ability to successfully expand in our business markets and into new markets and industry verticals; and |
| ● | our ability to effectively manage our growth and future expenses. |
Other risks and uncertainties include such factors, among others, as market acceptance and market demand for our products and services, pricing, the changing regulatory environment, the effect of our accounting policies, industry trends, adequacy of our financial resources to execute our business plan, our ability to attract, retain and motivate key personnel, and other risks described from time to time in periodic and current reports we file with the United States Securities and Exchange Commission, or the “SEC.” You should consider carefully the statements under this report, which address additional factors that could cause our actual results to differ from those set forth in the forward-looking statements and could materially and adversely affect our business, operating results and financial condition. All subsequent written and oral forward-looking statements attributable to us or persons acting on our behalf are expressly qualified in their entirety by the applicable cautionary statements.
| 3 |
PART I
ITEM 1. BUSINESS
Our Corporate History and Background
BullFrog AI Holdings, Inc. was incorporated in the State of Nevada in February 2020. BullFrog AI Holdings, Inc. is the parent company of BullFrog AI, Inc. and BullFrog AI Management, LLC, which were incorporated in Delaware and Maryland, in 2017 and 2021, respectively. All our operations are currently conducted through BullFrog AI Holdings, Inc. The Company’s principal business address is 325 Ellington Blvd, Unit 317, Gaithersburg, MD 20878. Our website address is www.bullfrogai.com. The references to our website in this Annual Report on Form 10-K (the “Form 10-K”) are inactive textual references only. The information on our website is not incorporated into this Form 10-K.
Acquisition of BullFrog AI, Inc.
In June 2020, BullFrog AI Holdings, Inc. acquired BullFrog AI, Inc. pursuant to an exchange agreement under which each share of BullFrog AI, Inc. common stock was exchanged for a share of common stock of the Company. Immediately prior to the share exchange, each outstanding common share of BullFrog AI, Inc. was split into 25 shares of common stock. Pursuant to the share exchange agreement, 24,223,975 shares of the Company’s common stock were issued to the shareholders of BullFrog AI, Inc. in exchange for 100% of the outstanding stock of BullFrog AI, Inc. Upon completion of the exchange, BullFrog AI, Inc. became the Company’s wholly-owned subsidiary and the shareholders of BullFrog AI, Inc. held 100% of the common stock of the Company. As a result, the Company assumed a total of $330,442 in net liabilities of BullFrog AI, Inc. Both of the entities were controlled before and after the transactions by the same controlling shareholder.
BullFrog AI Corporate History
BullFrog AI, Inc. was incorporated in the State of Delaware in August 2017. Vininder Singh, the Company’s Chief Executive Officer, was the founder, CEO and chairman of BullFrog AI, Inc.
Business Overview
Most new therapeutics will fail at some point in preclinical or clinical development. This is the primary driver of the high cost of developing new therapeutics. A major challenge in developing new therapeutics is efficiently integrating the complex, high-dimensional data generated at each stage of development to help de-risk subsequent stages of the process. Artificial intelligence and machine learning (“AI/ML”) has emerged as a digital solution to help address this problem.
We use AI/ML to advance medicines for both internal and external projects. We are committed to increasing the probability of success and decreasing the time and cost involved in developing therapeutics. Most current AI/ML platforms still fall short in their ability to synthesize disparate, high-dimensional data for actionable insight. Our platform technology, bfLEAP™, is an analytical AI/ML platform derived from technology developed at The Johns Hopkins University Applied Physics Laboratory (“JHU-APL”), which is able to surmount the challenges of scalability and flexibility currently hindering researchers and clinicians by providing a more precise1, multi-dimensional understanding of their data. We are deploying bfLEAP™ for use at several critical stages of development for internal programs and through strategic partnerships and collaborations with the intention of streamlining data analytics in therapeutics development, decreasing the overall development costs by decreasing failure rates for new therapeutics, and impacting the lives of countless patients that may otherwise not receive the therapies they need.
1 In an August 2021 publication in DeepAI.org (https://deepai.org/publication/random-subspace-mixture-models-for-interpretable-anomaly-detection), the algorithms used in bfLEAP were compared to 10 of the most popular clustering algorithms in the world using 12 data sets. The end result showed that the algorithms used in bfLEAP had the highest average score when measuring speed and accuracy of prediction. The bfLEAP platform currently has more advanced versions of these algorithms and is applying them in multiple data analytics projects.
| 4 |
Recent Developments
As previously reported, on August 21, 2025, the Company received written notice from The Nasdaq Stock Market LLC (“Nasdaq”) that the Company was not in compliance with Nasdaq Listing Rule 5550(b)(1), which requires companies listed on The Nasdaq Capital Market to maintain a minimum of $2,500,000 in stockholders’ equity for continued listing (the “Stockholders’ Equity Requirement”). The Company submitted a plan to regain compliance with the Stockholders’ Equity Requirement (the “Plan”) to Nasdaq on September 30, 2025. In response to the Company’s Plan, on October 7, 2025, the Company received a letter (the “Extension Letter”) from Nasdaq informing the Company that, based on the Plan, Nasdaq had granted the Company’s request for an extension until February 17, 2026, to comply with the Stockholders’ Equity Requirement. On February 19, 2026, the Company received a further notice from Nasdaq (the “February Letter”) notifying the Company that Nasdaq determined that the Company had not met the terms of the extension. The Company thereafter timely requested a hearing before an independent Nasdaq Hearings Panel (the “Panel”) that automatically stayed any suspension or delisting action pending the hearing and the expiration of any extension period granted by the Panel following the hearing. At the hearing, the Company plans to present additional details of the Company’s Plan and provide an update on its efforts to regain compliance. The Company will also request additional time to complete the steps of its Plan and regain compliance with all applicable Nasdaq Listing Rules. There can be no assurance that the Panel will grant the Company’s request for additional time to regain compliance with Nasdaq listing rules or that, if the Panel does grant the Company’s request, the Company will be able to regain compliance with the applicable Nasdaq listing requirements. If the Company’s common stock and warrants are delisted, it would be more difficult to buy or sell the Company’s common stock and warrants or to obtain accurate quotations, and the price of the Company’s common stock and warrants could suffer a material decline. Delisting could also impair the Company’s ability to raise capital.
On February 10, 2026, the Company received a letter from Nasdaq notifying the Company that, for the last 30 consecutive business days, the closing bid price for the Company’s common stock, par value $0.00001 per share (the “Common Stock”), was below $1.00 per share, which is the minimum closing bid price required for continued listing on the Nasdaq Global Market (the “Minimum Bid Price Requirement”) pursuant to Nasdaq Listing Rule 5550(a)(2) (the “Bid Price Notice”). The Bid Price Notice had no immediate effect on the listing of the Company’s Common Stock and tradeable warrants. As such, the Company’s Common Stock will continue to trade on the Nasdaq Capital Market under the symbol “BFRG,” and its tradeable warrants will continue to trade on the Nasdaq Capital Market under the symbol “BFRGW.” In accordance with Nasdaq Listing Rule 5810(c)(3)(A), the Company is provided a compliance period of 180 calendar days from the date of the Bid Price Notice, or until August 10, 2026, to regain compliance with the Minimum Bid Price Requirement. If at any time during the 180-calendar day grace period, the closing bid price of the Company’s Common Stock is at least $1.00 per share for a minimum of ten consecutive business days (unless the Nasdaq staff exercises its discretion to extend this ten business day period pursuant to Nasdaq Listing Rule 5810(c)(3)(H)), Nasdaq will provide the Company written confirmation of compliance, and the matter will be closed. If the Company does not regain compliance during the initial 180-calendar day compliance period, the Company may be provided a second 180-calendar day period to regain compliance. If the Company does not regain compliance within the allotted compliance periods, including any extensions that may be granted by Nasdaq, the Company’s listed securities will be subject to delisting. The Company would thereafter have the right to appeal a determination to delist the Company’s securities, and the Company’s securities would remain listed on the Nasdaq Capital Market until the completion of the appeal process. The Company intends to monitor the closing bid price of its Common Stock and assess potential options to regain compliance with Nasdaq’s Listing Rules. While the Company plans to review all available options, there can be no assurance that the Company will regain compliance with the Minimum Bid Price Requirement during the compliance period, secure a second 180-day period to regain compliance with the Minimum Bid Price Requirement, or maintain compliance with the other Nasdaq listing requirements. Notably, at a Special Meeting of Stockholders in October 2025, the Company received stockholder approval to effect a reverse stock split at a ratio of not less than 1-to-2 and not more than 1-to-15, such ratio and timing to be determined in the discretion of the Company’s Board of Directors.
Our Strategy
We plan to achieve our business objectives by enabling the successful discovery and/or development of drugs and biologics using a precision medicine approach via our proprietary artificial intelligence platform bfLEAP™. The bfLEAP™ platform utilizes both supervised and unsupervised machine learning; as such, it is able to reveal meaningful connections in the data without the need for a priori hypothesis. Supervised machine learning uses labeled input and output data, while an unsupervised learning algorithm does not. In supervised learning, the algorithm “learns” from the training dataset by iteratively making predictions on the data and adjusting for the correct answer. Unsupervised learning, also known as unsupervised machine learning, uses machine learning algorithms to analyze and cluster unlabeled datasets. These algorithms discover hidden patterns or data groupings without the need for human intervention. Algorithms used in the bfLEAP™ platform are designed to handle highly imbalanced data sets to successfully identify combinations of factors that are associated with outcomes of interest.
| 5 |
Together with our strategic partners and collaborators, our primary goal is to improve the odds of success at any stage of pre-clinical and clinical therapeutics development. Our primary business model is improving the success and efficiency of drug development which is accomplished either through acquisition of drugs or partnerships and collaborations with companies that are developing drugs. We hope to accomplish this through strategic acquisitions of current clinical stage and failed drugs for in-house development, or through strategic partnerships with biopharmaceutical industry companies. We are able to pursue our drug asset enhancement business by leveraging a powerful and proven AI/ML platform, bfLEAP™, initially derived from technology developed at JHU-APL. We believe the bfLEAP™ analytics platform is a potentially disruptive tool for analysis of pre-clinical and clinical data sets, such as the robust pre-clinical and clinical trial data sets being generated in translational R&D and clinical trial settings. In November 2021, we amended our license agreement with JHU-APL to include additional advanced AI technology. In July 2022, the Company entered into an exclusive, world-wide, royalty-bearing license from JHU-APL for the additional technology developed to enhance the bfLEAP™ platform. The July 2022 JHU-APL license provides the Company with new intellectual property and also encompasses most of the intellectual property from our original February 2018 license agreement with JHU-APL.
We believe bfLEAP™ will inform and enable decision making throughout the development cycle:
| ● | Discovery Phase – Analyze and categorize discovery phase data to better define highest-value leads from groups of candidates, for advancement to preclinical phase of development. Integrate data from high-throughput screening, pharmacodynamics assays, pharmacokinetics assays, and other key data sets to create the most accurate profile of a pool of therapeutic candidates. There is often a high degree of similarity among closely related therapeutics in a candidate pool; bfLEAP™ is able to harmonize disparate data streams for a more nuanced understanding of each candidate’s characteristics and potency. |
| ● | Pre-Clinical Data – Large-scale, multivariate analysis of pre-clinical and early-stage clinical data sets. In these settings, bfLEAP™ could be used to find novel drug targets, elucidate mechanism of action, predict potential off-target effects and side effects, uncover specific genetic and phenotypic backgrounds with highest correlation to therapeutic response, etc. These insights from bfLEAP™ analysis can be used to inform decision making and study design at the subsequent steps of therapeutic and diagnostic development, including first-inhuman and Phase I randomized controlled trials. |
| ● | Clinical Development – Advanced, multivariate analysis of Phase I and Phase II clinical trials data, to find niche populations of highly responsive patients and/or inform patient selection for later-stage clinical trials. This can be used to decrease overall study risk for larger clinical trials, including Phase II trials, and any Phase III Registration Clinical Trials. The bfLEAP™ platform analysis can also be used to more precisely understand complex correlations between therapeutic treatment and adverse events, side effects, and other undesirable responses which could jeopardize clinical trial success. |
Our platform is agnostic to the disease indication or treatment modality and therefore we believe that it is of value in the development of biologics or small molecules.
The process for our drug asset enhancement program is to:
| ● | acquire the rights to a drug from a biopharmaceutical industry company or academia; |
| ● | use the proprietary bfLEAP™ AI/ML platform to determine a multi-factorial profile for a patient that would best respond to the drug; |
| ● | rapidly conduct a clinical trial to validate the drug’s use for the defined “high-responder” population; and |
| ● | divest and sell the rescued drug asset with the new information back to a large player in the pharma industry, following positive results of the clinical trial. |
| 6 |
As part of our strategy, we will continue evolving our intellectual property, analytical platform and technologies, build a large portfolio of drug candidates, and implement a model that reduces risk and increases the frequency of cash flow from rescued drugs. This strategy will include strategic partnerships, collaborations, and relationships along the entire drug development value chain, as well as acquisitions of the rights to developing failed drugs and possibly the underlying companies.
To date, we have not conducted clinical trials on any pharmaceutical drugs and our platform has not been used to identify a drug candidate that has received regulatory approval for commercialization. However, we currently have a strategic relationship with a leading rare disease non-profit organization for AI/ML analysis of late-stage clinical data. We have acquired the rights to a series of preclinical and early clinical drug assets from universities and entered into a strategic collaboration with a world-renowned research institution to create a HSV1 viral therapeutic platform to engineer immunotherapies for colorectal cancer. We have signed exclusive worldwide license agreements with Johns Hopkins University for a cancer drug that targets glioblastoma (brain cancer), pancreatic cancer, and other cancers. We have also signed an exclusive worldwide license with George Washington University for another cancer drug that targets hepatocellular carcinoma (liver cancer), and other liver diseases.
Our platform was originally developed by JHU-APL. JHU-APL uses the same technology for applications related to national defense. Over several years, the software and algorithms have been used to identify relationships, patterns, and anomalies, and make predictions that otherwise may not be found. These discoveries and insights provide an advantage when predicting a target of interest, regardless of industry or sector. We have applied the technology to various clinical data sets and have identified novel relationships that may provide new intellectual property, new drug targets, and other valuable information that may help with patient stratification for a clinical trial, thereby improving the odds for success. The platform has not yet aided in the development of a drug that has reached commercialization; however, we have licensed one drug candidate that has completed a Phase I trial and a second candidate that is in the preclinical stages. Our aim is to use our technology on current and future available data to help us better determine the optimal path for development.
While we have not generated significant revenues from our AI/ML operations, we anticipate generating revenue in the future from the following three sources:
Contract Services
Our fee for service partnership offering model is designed for biopharmaceutical companies, as well as other organizations of all sizes that have challenges analyzing data throughout the drug development process. We provide the customer with an analysis of large complex data sets using our proprietary AI/ML platform called bfLEAP™. This platform is designed to predict targets of interest, patterns, relationships, and anomalies. Our service model involves fees in cash and, in some instances, equity or other consideration and, in some instances, the potential for rights to new intellectual property generated from the analysis, which can be performed at the discovery, preclinical, or clinical stages of drug development.
Collaborative Arrangements
We plan to enter into collaborative arrangements with biotechnology and pharmaceutical companies who have drugs that are in development or have failed late Phase II or Phase III trials. The collaborations may also be at the discovery or preclinical stages of drug development. Our revenue will be a combination of fee for service payments and success fees based on achieving certain milestones as determined by each specific arrangement. There may also be fees or legal rights associated with the development of new intellectual property.
Acquisition of Rights to Certain Drugs
We may acquire the rights to drugs that have failed late Phase II or Phase III trials and generate revenues by using our platform to accurately determine the profile of patients that would respond to the drugs, conduct a clinical trial to test our findings either independently or with a clinical partner, and finally sell the drug back to pharmaceutical companies. We have and may continue acquiring the rights to drugs that have not yet failed any trials. We will use our technology to improve the chances for success, conduct a trial, and divest the asset. When divesting assets, the transaction may involve a combination of upfront payments, milestone payments based on clinical success, and royalties on sales of the product.
| 7 |
Our Products
| Product/Platform | Description | Target Market/Indications | ||
| bfLEAP™ – AI/ML platform for analysis of preclinical and clinical data | AI/ML analytics platform derived from technology developed at JHU-APL and licensed by the Company. | Biotechnology and pharmaceutical companies and other organizations. | ||
| BullFrog Data Networks™ | AI-powered bioinformatics solution that integrates and analyzes multi-omics, clinical, and real-world biomedical data to uncover hidden biological relationships and generate insights that accelerate drug discovery, target identification, and clinical trial design. | Biotechnology and pharmaceutical companies and other organizations. | ||
| bfPREP™ | AI-powered data preparation platform that cleans, standardizes, and harmonizes fragmented biomedical datasets (e.g., clinical, omics, and real-world data) to convert them into analysis-ready, AI-ready data for drug discovery and clinical research. | Biotechnology and pharmaceutical companies and other organizations. | ||
| siRNA | Small interfering RNA targeting Beta2-spectrin in the treatment of human diseases developed at George Washington University and licensed by the Company. Product has not yet initiated clinical testing. | Hepatocellular carcinoma, treatment of obesity, non-alcoholic fatty liver disease, and non-alcoholic steatohepatitis. | ||
| Mebendazole | Improved formulation of Mebendazole developed at Johns Hopkins University and licensed by the Company. Product has begun the process of clinical testing but has not received regulatory approval for commercialization. | Glioblastoma. |
In January 2022, we entered into an exclusive, worldwide, royalty-bearing license from George Washington University (“GWU”) for rights to use siRNA targeting Beta2-spectrin in the treatment of human diseases, including hepatocellular carcinoma (“HCC”). The license covers methods claimed in three U.S. and worldwide patent applications, and also includes use of this approach for treatment of obesity, non-alcoholic fatty liver disease, and non-alcoholic steatohepatitis. This program is currently in the preclinical stage of development. We initiated proof-of-concept studies on this asset and plan to use the outcome of these studies to inform a clinical development plan that would include initiation of IND-enabling studies.
Metabolic dysfunction-associated steatotic liver disease (known as MASLD, which until recently was called non-alcoholic fatty liver disease, or NAFLD) is a condition in which excess lipids, or fat, build up in the liver. This condition, which is more common in people who have obesity and related metabolic diseases including type 2 diabetes, affects as many as 24% of adults in the United States and is associated with risk of progression to more serious conditions, including metabolic dysfunction-associated steatohepatitis (“MASH”), with associated liver inflammation and fibrosis, and HCC. Evidence in animal models of obesity suggest that a protein called β2-spectrin may play a key role in lipid accumulation, tissue fibrosis, and liver damage, and targeting expression or activity of this protein may be a useful approach in treating MASH and liver cancer (Rao et al., 2021).
| 8 |
In February 2022, we entered into an exclusive, worldwide, royalty-bearing license with Johns Hopkins University (“JHU”) for the use of an improved formulation of Mebendazole for the treatment of any human cancer or neoplastic disease. This formulation shows potent activity in animal models with different types of cancer and has been evaluated in a Phase I clinical trial in patients with high-grade glioma (NCT01729260). The trial, an open-label dose-escalation study, assessed the safety of the improved formulation with adjuvant temozolomide in 24 patients with newly diagnosed gliomas. Investigators observed no dose-limiting toxicity in patients receiving all but the highest tested dose (200mg/kg/day). Four of the 15 patients receiving the maximum tested dose of 200mg/kg/day experienced dose-limiting toxicity, all of which were reversed by decreasing or eliminating the dose given. There were no serious adverse events attributed to mebendazole at any dose during the trial. We are currently formulating a strategy to find a partner to conduct additional clinical trials with this asset to enable evaluation of safety in humans.
In October 2022, we entered into an exclusive, world-wide, royalty-bearing license with JHU and the Institute of Organic Chemistry and Biochemistry (“IOCB”) of the Czech Academy of Sciences for rights to commercialize N-substituted prodrugs of mebendazole that demonstrate improved solubility and bioavailability. The license covers prodrug compositions and use for treating disease as claimed in multiple United States and worldwide patent applications. Patents have since been issued in the United States and Australia and are still in the prosecution phase in other territories. In September 2023, the Company announced results from a preclinical study demonstrating the effectiveness of BF-223, a compound chosen from this class, in an animal model for glioblastoma. The Company is currently formulating a strategy for initiating IND-enabling studies on BF-223 and is conducting outreach to identify partners that may want to license or partner in the development of BF-223.
Our bfLEAP™ Analytics Platform
We are able to pursue our drug rescue business by leveraging bfLEAP™, our powerful and proven AI/ML platform derived from technology developed at JHU-APL. The bfLEAP™ platform is based on an exclusive, world-wide license granted by JHU-APL. The license covers three (3) issued patents, as well as a new provisional patent application, non-patent rights to proprietary libraries of algorithms and other trade secrets, which also includes modifications and improvements. In July 2022, we entered into an exclusive, world-wide, royalty-bearing license from JHU-APL for the additional technology developed to enhance the bfLEAP™ platform, which superseded our original license with JHU-APL. This license provides additional intellectual property rights including patents, copyrights and knowhow to be utilized under our bfLEAP™ analytical AI/ML platform. Under the terms of such license agreement, JHU-APL will be entitled to eight percent (8%) of net sales for the services provided by the Company to other parties and three percent (3%) for internally developed drug projects in which the JHU-APL license was utilized. The license also contains tiered sub licensing fees that start at 50% and reduce to 25% based on revenues.
We believe the bfLEAP™ analytics platform is a potentially disruptive tool for analysis of pre-clinical and clinical data sets, such as the robust pre-clinical and clinical trial data sets being generated in translational R&D and clinical trial settings. The input data for bfLEAP™ can include raw data (preclinical and/or clinical readouts), categorical data, sociodemographic data of patients, and various other inputs. Thus, the bfLEAP™ platform is capable of capturing the particular genetic and physical characteristics of patients in an unbiased manner, and contextualizing it against other disparate data sources from patients (e.g. molecular data, physiological data, etc.) for less biased and more meaningful conclusions. It is also uniquely scalable; the bfLEAP™ platform is able to perform analysis on large, high-volume data sets (i.e. ‘big data’) and also able to analyze highly disparate “short and wide” data as well. In terms of visualization, bfLEAP™ is able to integrate with most commonly used visualization tools for graph analytics.
We believe that the combination of (a) scalable analytics (i.e., large data or short/wide data), (b) state-of-the-art proprietary algorithms, (c) unsupervised machine learning, and (d) streamlined data ingestion and visualization makes bfLEAP™ one of the most flexible and powerful new platforms available on the market.
We intend to continue to evolve and improve bfLEAP™.
BullFrog Data Networks™
The BullFrog Data Networks™ solution incorporates publicly available, proprietary, and custom data sources to generate novel insights toward target identification and validation, understanding mechanism of action, clinical trial optimization, drug repurposing, and more. BullFrog Data Networks™ can be created for any therapeutic area, and we use this technology in our own research and development pipeline including in oncology and central nervous system indications. Our approach finds patient subgroups with similar molecular signatures and identifies the most relevant genes driving disease biology. We believe that data networks created from this process generate insights that allow researchers to accelerate drug discovery and development and increase the odds of technical and regulatory success. In February 2025, we entered into a collaboration agreement with Eleison Pharmaceuticals Inc. (“Eleison”), a Phase III oncology company focused on novel chemotherapeutic treatments for rare cancers, where we applied our proprietary BullFrog Data Networks™ solution, powered by the bfLEAP® platform, and we plan to offer this solution to other biotechnology and pharmaceutical companies going forward.
| 9 |
bfPREP™
The bfPREP™ solution is an AI-powered data preparation tool designed to organize and standardize complex biomedical datasets before advanced analytics or modeling. It ingests heterogeneous data sources such as clinical trial records, multi-omics datasets, and real-world patient data and automatically cleans, harmonizes, and structures them into consistent, analysis-ready formats. The platform reduces the time and manual effort researchers typically spend on data curation, which can be a major bottleneck in biomedical research. By preparing high-quality, integrated datasets, bfPREPTM helps ensure that downstream AI models and analytical tools produce more reliable and reproducible insights. It is designed to work as part of the company’s broader BullFrog Data NetworksTM ecosystem, supporting applications such as biomarker discovery, drug target identification, and clinical trial optimization. We believe that bfPREPTM enables life sciences organizations to move from fragmented raw data to AI-ready datasets much more efficiently.
New Scenario-Based Decision Engine
In late March 2026, the Company is planning to launch a new scenario-based decision engine solution that is an AI-powered analytical environment designed to support complex decision-making in drug development and biomedical research. The platform will apply advanced modeling and simulation techniques to evaluate multiple development scenarios, allowing researchers and executives to explore the potential outcomes of different clinical, regulatory, and commercialization strategies. By integrating diverse datasets including biological, clinical, and operational information, the new scenario-based decision engine will help identify which approaches are most likely to succeed while highlighting potential risks. The new scenario-based decision engine is intended to support portfolio prioritization, clinical trial design, and strategic planning by providing quantitative insight into uncertain R&D decisions. It will operate alongside the Company’s broader BullFrog Data NetworksTM platform, which supplies the integrated data foundation for its analyses. Together, we believe that these capabilities can help life sciences organizations make more informed, data-driven decisions throughout the drug development process.
Lieber Institute for Brain Development
In September 2023, the Company entered a data use and technology partnership agreement (the “Partnership Agreement”) with the Lieber Institute for Brain Development (“LIBD”). The Partnership Agreement covers the right of the Company to leverage its bfLEAP™ platform to mine LIBD’s comprehensive brain data, including transcriptomic, genomic, DNA methylation, cell-line, clinical, and imaging data to identify previously unrecognized relationships. The goal of the partnership is to identify previously unrecognized relationships between genes and pathways in the brain and the development of neurologic and psychiatric disorders, thereby facilitating the development of more effective treatments for diseases of the human brain. The collaboration will proceed in two stages, with the first involving unsupervised construction of graphical models to reveal relationships between brain diseases and genomic/biologic attributes, with the goal of identifying new biomarkers and drug targets across disorders. The second stage will involve creating disease-specific models that will enable identification of genes and pathways within these respective disorders. The Partnership Agreement had a one-year term of data exclusivity to complete the first stages of analyses, with a two-year extension option as performance milestones are met.
As contemplated in the Partnership Agreement, in October 2023, we entered into a commercial agreement (the “Commercial Agreement”) with LIBD that sets forth the key terms for commercialization of products and services developed under the Partnership Agreement. Pursuant to the Commercial Agreement, LIBD granted the Company a worldwide, royalty-bearing exclusive license so long as the Company receives net sales or income from the licensing of “Licensed Products” (as defined in the Commercial Agreement) in the application of machine learning and artificial intelligence for research and development in drug development, and specifically includes therapeutic products, patient selection strategies, and target identification, but excludes diagnostics and incidental uses of machine learning and/or artificial intelligence on data derived from research. Generally, “Licensed Products” are any product or service which incorporates, results from, or is derived from LIBD’s Data (meaning finished brain-related data, including but not limited to DNA methylation, RNAseq, genomic, DNA methylation, cell-line, clinical, and imaging data, and the specified data set forth in the Partnership Agreement) and that we or our affiliate develops during the term of the Partnership Agreement, and any improvements thereof after the term of the Partnership Agreement, and all Licensed Products or services derived therefrom by us or our affiliates. Licensed Products may include, but are not limited to, biomarker and target identification, target validation, mapping unmet needs, identifying genetic risk factors and predictive modeling.
| 10 |
We were also granted the right to sublicense, to use the deliverables under the Partnership Agreement, and LIBD’s intellectual property rights in the data, to (i) use, sell, distribute for sale, have distributed for sale, offer for sale, have sold, import and have imported Licensed Products and (ii) to develop, have developed, make, have made Licensed Products that are derived from Licensed Products developed during the term of the Partnership Agreement, and any improvements made following the term. We are prohibited from sublicensing LIBD Data. We shall pay LIBD a royalty based on net sales of all Licensed Products sold by the Company and its affiliates.
The Commercial Agreement, generally, may be terminated at any time by either us or LIBD if either party defaults or breaches any material term of the agreement or files for protection under bankruptcy laws, makes an assignment for the benefit of creditors, or appoints or suffers appointment of a receiver, trustee, or similar agent over its property.
Summary for CATIE Schizophrenia Case Study
As part of the Partnership Agreement, the Company worked with LIBD to analyze data from the landmark Clinical Antipsychotic Trials of Intervention Effectiveness (“CATIE”) trials. The CATIE trials were the largest trials ever conducted for anti-psychotic medications. The Company analyzed CATIE data from ~200 schizophrenia patients, with a library of almost 1 million genetic data points for each patient, more than 200 non-genetic attributes per patient, and 4 different medications used in the trial. For each of the four medications used, bfLEAP™ analysis revealed new, previously unknown relationships between individual genetic variants and negative patient symptoms. The genetic loci identified represent potential druggable targets, as well as potential stratifying criteria for future clinical trials in schizophrenia.
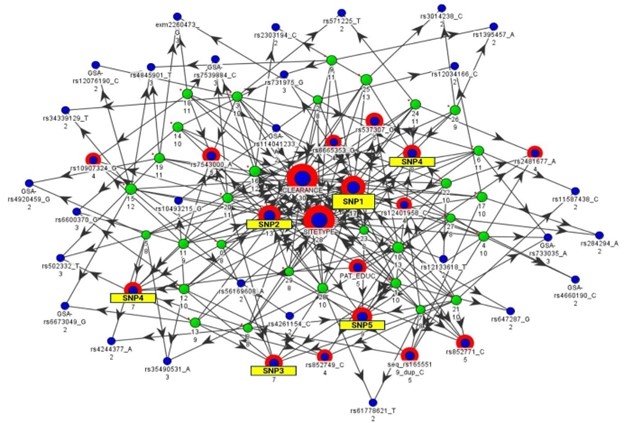
We performed another analysis on the data using our new advanced clustering algorithms, bfLEAP 2.0, but focused on one particular drug named Olanzapine. Our bfLEAP™ 2.0 analytical results identified previously unknown, multi-dimensional associations among newly identified genetic variants, drug clearance, clinical trial sites, and clinical outcome variables in schizophrenia patients.

FIGURE 1 – bfLEAP™ Analytical Map
| 11 |
Each green node represents a different sampling of the data, and arrows point to attributes (blue nodes) which were found to be key indicators according to that sampling. Attribute importance is determined by how many samplings identify that attribute as an indicator (i.e., number of incoming arrows to each blue node).
Identification of clustered multi-variate associations (e.g., novel genetic variants, drug clearance, substance abuse) could help us (1) identify novel drug targets, (2) predict which patients are most likely to respond, and (3) identify modifiable factors that could contribute to better outcomes.
Summary for Cardiovascular Case Study
We worked with an international collaborator in cardiovascular devices to analyze data from an ongoing clinical trial for a new device. BullFrog analyzed data from ~55 patients, with a library of almost 15,000 unique attributes of data for each patient. The data also included adverse events and key demographic information. For this collaborator, bfLEAP™ analysis was able to provide ground truth, confirming multiple correlations and non-correlations within the data. In terms of actionable output, the analytical results confirmed at least two demographic co-variates for the ongoing trial, and also provided a starting point for deeper physiological and molecular studies.
Our Supply Chain and Customer Base
We launched our businesses using funds from our initial public offering and we continue operating and targeting partnerships and relationships using funds from our subsequent financings. We have a strategic relationship with FSHD Society, a leading non-governmental organization, for AI/ML analysis of clinical trial data for patients with a rare neuromuscular disorder. We also have several other developing strategic relationships in the project design phase. The Company has executed a joint development deal for a biologics discovery phase opportunity that is directed toward targeted cancer therapeutics. The Company has also obtained exclusive worldwide rights to a Phase II ready glioblastoma drug and a discovery phase hepatocellular carcinoma drug from universities. Since we intend to conduct late-stage clinical trials with partners on rescued therapeutic assets, there will be a requirement of drug product or other significant services to plan and execute our clinical development programs. The success of our partnered clinical development programs will require adequate availability of raw materials and drug product for our R&D and clinical trials, and, in some cases, may also require establishment of third-party arrangements to obtain finished drug product that is manufactured appropriately under industry-standard guidelines, and packaged for clinical use or sale. Since we are a digital biopharmaceutical company, our clinical development programs will also require, in some cases, the establishment of third-party relationships for execution and completion of clinical trials.
| 12 |
Our Market Opportunity
One aim of our business is to “rescue” drugs that have failed in Phase III clinical trials by using our technology to analyze all available data with the goal of designing a precision medicine clinical trial that will have a better chance of being successful. The graphic below illustrates the estimated market opportunity for these failed drugs. The top arrow shows the number of failed Phase III trials for several disease categories over a 5-year period. The arrows below provide our assumptions for narrowing or discounting certain parameters associated with the market size calculation. The final arrow shows the math behind the $47.1 billion market opportunity. To date, we have not penetrated the failed drug market, however; we are actively searching for failed drug opportunities.

Identification of candidates with potential for rescue may be challenging and require significant resources, and once these assets are identified, we may find it challenging to license them under favorable terms in order to create value for shareholders. Subsequent development of these assets for clinical testing may require significant effort and resources. Ultimately, these assets must undergo rigorous clinical testing and approval by FDA or comparable regulatory authorities in other countries in order to be marketed. A key part of our strategy is to partner our R&D programs. In addition, we do not intend on commercializing drugs and instead will seek to divest each drug asset to a company that will commercialize the drug. We may receive future royalties in some transactions.
| 13 |
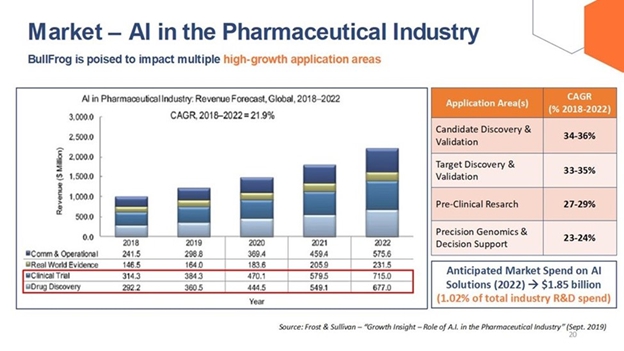
The following graphic illustrates the global revenue forecast for applying AI in the pharmaceutical industry, as well as the increase in anticipated market spend and annual growth rate for AI solutions per certain application areas.
Intellectual Property
Patents
We have exclusive worldwide rights to the following patents related to our intellectual property:
Mebendazole Polymorph For Treatment And Prevention Of Tumors
| Serial Number | Country | Status | Issue Date | Expiration Date | ||||
| 62/112,706 | United States | Converted | N/A | N/A | ||||
| PCT/US2016/016968 | PCT | Nationalized | N/A | N/A | ||||
| 11,110,079 | United States | Granted | 9/7/2021 | 2/8/2036 | ||||
| 17/402,131 | United States | Abandoned | N/A | N/A | ||||
| 18/525,209 | United States | Pending | N/A | N/A | ||||
| 16747414.7 | Europe | Granted | 12/15/2021 | 2/8/2036 | ||||
| 16747414.7 | Czech Republic | Granted | 12/15/2021 | 2/8/2036 | ||||
| 16747414.7 | France | Granted | 12/15/2021 | 2/8/2036 | ||||
| 60 2016 067 384.3 | Germany | Granted | 12/15/2021 | 2/8/2036 | ||||
| 16747414.7 | Ireland | Granted | 12/15/2021 | 2/8/2036 | ||||
| 502022000018341 | Italy | Granted | 12/15/2021 | 2/8/2036 | ||||
| 16747414.7 | Spain | Granted | 12/15/2021 | 2/8/2036 | ||||
| 16747414.7 | Switzerland | Granted | 12/15/2021 | 2/8/2036 | ||||
| 16747414.7 | United Kingdom | Granted | 12/15/2021 | 2/8/2036 | ||||
| 253854 | Israel | Granted | 6/26/2021 | 2/8/2036 | ||||
| 2016800144274 | China | Granted | 6/25/2021 | 2/8/2036 | ||||
| 201717028684 | India | Granted | 12/1/2020 | 2/8/2036 | ||||
| 2017-541687 | Japan | Granted | 11/18/2020 | 2/8/2036 |
Mebendazole Prodrugs with Enhanced Solubility and Oral Bioavailability
| Serial Number | Country | Status | Issue Date | Expiration Date | ||||
| 62/627,810 | United States | Converted | N/A | N/A | ||||
| PCT/US2019/017291 | PCT | Nationalized | N/A | N/A | ||||
| 11,712,435 | United States | Granted | 8/1/2023 | 2/8/2039 | ||||
| 2019216757 | Australia | Granted | 1/4/2024 | 2/8/2039 | ||||
| 19751700.6 | Europe | Pending | N/A | N/A | ||||
| 3,090,691 | Canada | Pending | N/A | N/A |
| 14 |
Inhibition of SPTBN1 to treat Obesity/NASH and Obesity/NASH-driven cancer
| Serial Number | Country | Status | Filing Date | Expiration Date | ||||
| 63/113,745 | United States | Converted | 11/13/2020 | N/A | ||||
| 63/147,141 | United States | Converted | 2/8/2021 | N/A | ||||
| PCT/US2021/059245 | United States | Nationalized | 11/12/2021 | N/A | ||||
| 2023-528428 | Japan | Filed | 11/12/2021 | N/A | ||||
| 18/252,771 | United States | Filed | 5/12/2023 | N/A | ||||
| 21892928.9 | Europe | Filed | 6/13/2023 | N/A | ||||
| 2021800763877 | Canada | Filed | 11/12/2021 | N/A |
Johns Hopkins University Applied Physics Lab Licensed Intellectual Property
| Title | Serial Number | File Date | Country | Status | Expiration Date | Assignee | ||||||
| Apparatus and Method for Distributed Graph Processing | U.S. Patent 10,146,801 | 7/13/2015 | US | Granted | 3/2/2037 | The Johns Hopkins University | ||||||
| Method and Apparatus for Analysis and Classification of High Dimensional Data Sets | U.S. Patent 10,936,965 | 10/5/2017 | US | Granted | 9/25/2038 | The Johns Hopkins University | ||||||
| Generalized Low Entropy Mixture Model | U.S. Patent 10,839,256 | 4/2/2018 | US | Granted | 12/15/2038 | The Johns Hopkins University |
Licenses
We hold the following licenses related to our intellectual property:
| Licensor | Licensee | Description of Rights Granted | ||
| Johns Hopkins University Applied Physics Lab | BullFrog AI, Inc. | Worldwide, exclusive rights for therapeutics development and analytical services | ||
| George Washington University | BullFrog AI Holdings | Worldwide, exclusive rights for therapeutics development | ||
| Johns Hopkins University | BullFrog AI Holdings | Worldwide, exclusive rights for therapeutics development |
JHU-APL Technology License
In February 2018, we entered into an exclusive, world-wide, royalty-bearing license with JHU-APL (the “2018 License Agreement”). The license covers three (3) issued patents, one (1) new provisional patent application, non-patent rights to proprietary libraries of algorithms and other trade secrets, as well as modifications and improvements. In October 2021, we executed an amendment to the original license for improvements and new advanced analytics capabilities. In consideration of the rights granted to the Company under the 2018 License Agreement, JHU-APL received a warrant equal to five percent (5%) of the then fully diluted equity base of the Company, which was diluted following the closing of our initial public offering. Under the terms of the 2018 License Agreement, JHU-APL will be entitled to an eight percent (8%) royalty on net sales for the services provided by us as well as fifty percent (50%) of all sublicense revenues received by us on services and sublicenses in which the JHU-APL licensed technology was utilized. In addition, we are required to pay JHU-APL an annual maintenance fee of $1,500. Minimum annual royalty payments are $20,000 for 2022, $80,000 for 2023, and $300,000 per year for 2024 and beyond. If cumulative annual royalty payments do not reach these levels, the amount due to JHU-APL to reach the annual minimum is due by January 1st of the following year. Failure to make annual royalty payments is considered a material breach under the agreement and, upon notice from JHU-APL of a material breach, we will have 60 days to cure the material breach.
| 15 |
In July 2022, the Company entered into an exclusive, world-wide, royalty-bearing license from JHU-APL for the additional technology developed to enhance the bfLEAP™ platform (the “2022 License Agreement”). The 2022 License Agreement provides additional intellectual property rights including patents, copyrights, and know-how to be utilized under the bfLEAP™ analytical AI/ML platform. This 2022 License Agreement supersedes the previous 2018 License Agreement. In consideration of the new license, we issued 39,879 shares of common stock to JHU-APL. Under the terms of the 2022 License Agreement, JHU-APL will be entitled to eight percent (8%) of net sales for the services provided by us to other parties and three percent (3%) for internally developed drug projects in which the JHU-APL license is utilized. The license also contains tiered sub licensing fees that start at 50% and reduce to 25% based on revenues. In addition, under the 2022 License Agreement, the minimum annual royalty payments are $30,000 for 2022, $80,000 for 2023, and $300,000 per year for 2024 and beyond, all of which are creditable by royalties. The financial terms of the new license agreement replaces the original terms from the 2018 License Agreement and are not duplicative.
In May 2023, we entered into Amendment Number 1 of the July 2022 License Agreement with JHU-APL whereby we gained access to certain improvements including additional patents and know-how in exchange for a series of payments totaling $275,000. The first of these payments for $75,000 was paid in July 2023, the second of these payments for $75,000 was paid in June 2025, and the remaining payments of $75,000 and $50,000 are due in 2026 and 2027, respectively. The amendment also reduced the 2023 minimum annual royalty payment from $80,000 to $60,000 while all other financial terms remained the same.
George Washington University - Beta2-spectrin siRNA License
In January 2022, we entered into an exclusive, world-wide, royalty-bearing license from GWU for rights to use siRNA targeting Beta2-spectrin in the treatment of human diseases, including hepatocellular carcinoma (“HCC”). The license covers methods claimed in three (3) U.S. and worldwide patent applications, and also includes use of this approach for treatment of obesity, non-alcoholic fatty liver disease, and non-alcoholic steatohepatitis. This program is currently in the preclinical stage of development and we have not yet initiated development activities or IND-enabling studies on this asset. All R&D on this candidate to date has been conducted by the licensor of the technology, GWU. The term of the agreement began in January 2022 and ends on the expiration date of the last patent to expire or 10 years after the first sale of a licensed product if no patents have been issued. The license can be terminated by the licensee upon 60 days’ written notice, or by the licensor if we are more than 30 days late in paying amounts owed to the licensor and do not make payment upon demand, or in the event of any material breach of the license that is not cured within 45 days.
Non-alcoholic fatty liver disease (“NAFLD”) is a condition in which excess lipids, or fat, build up in the liver. This condition, which is more common in people who have obesity and related metabolic diseases including type 2 diabetes, affects as many as 24% of adults in the United States and is associated with risk of progression to more serious conditions, including non-alcoholic steatohepatitis (“NASH”), with associated liver inflammation and fibrosis, and hepatocellular carcinoma (“HCC”). Evidence in animal models of obesity suggest that a protein called β2-spectrin may play a key role in lipid accumulation, tissue fibrosis, and liver damage, and targeting expression or activity of this protein may be a useful approach in treating NASH and liver cancer (Rao et al., 2021).
In consideration of the rights granted to us under the license agreement, GWU received a $20,000 license initiation fee. Under the terms of the license agreement, GWU will be entitled to a three percent (3%) royalty on net sales subject to quarterly minimums once the first sale has occurred subsequent to regulatory approval, as well sublicense or assignment fees in the event we sublicense or assign our rights to use the technology. We will also reimburse GWU for previously incurred and ongoing patent costs. The sublicense and assignment fee amounts decline as the Company advances the clinical development of the licensed technology. The license agreement also contains milestone payments for clinical development through the approval of a New Drug Application (“NDA”) and commercialization.
Aggregate future milestone costs could reach $860,000 if the drug successfully completes clinical trials and is the subject of an NDA to the United States FDA. Future milestones on sales revenue are limited to $1 million on the first $20 million in net sales.
| 16 |
We assessed whether the license should be capitalized and determined that the licensed program is in the early stage and therefore may not be recoverable; we expensed the license fee and will expense development costs until commercial viability is likely.
Johns Hopkins University – Mebendazole License
In February 2022, we entered into an exclusive, worldwide, royalty-bearing license with JHU for the use of an improved formulation of Mebendazole for the treatment of any human cancer or neoplastic disease. This formulation shows potent activity in animal models with different types of cancer, and has been evaluated in a Phase I clinical trial in patients with high-grade glioma (NCT01729260). The trial, an open-label dose-escalation study, assessed the safety of the improved formulation with adjuvant temozolomide in 24 patients with newly diagnosed gliomas. Investigators observed no dose-limiting toxicity in patients receiving all but the highest tested dose (200mg/kg/day). Four of the 15 patients receiving the maximum tested dose of 200mg/kg/day experienced dose-limiting toxicity, all of which were reversed by decreasing or eliminating the dose given. There were no serious adverse events attributed to mebendazole at any dose during the trial. We are currently formulating a strategy to conduct additional clinical trials with this asset to enable evaluation of safety in humans.
The license covers six (6) issued patents and one (1) pending application, with the term of the agreement beginning in February 2022 and ending on the date of expiration of the last to expire patent. The license can be terminated by the licensee upon 90 days’ written notice, or by the licensor in the event of any material breach of the license that is not cured within 30 days. In consideration of the rights granted to us under the license agreement, JHU received a staggered upfront license fee of $250,000, with the first $50,000 paid in 2022 and the remaining balance of $200,000 paid in 2023. The Company also reimbursed JHU for previously incurred and ongoing patent costs. Under the terms of the license agreement, JHU will be entitled to three- and one-half percent (3.5%) royalty on net sales in which the JHU license was utilized. In addition, we are required to pay JHU minimum annual royalty payments of $5,000 for 2022, $10,000 for 2023, $20,000 for 2024, $30,000 for 2025 and $50,000 for 2026 and each year after until the first commercial sale, after which the annual minimum royalty shall be $250,000. The license agreement also contains milestone payments for clinical development steps through the approval of an NDA and commercialization. Aggregate future milestone costs could reach $1,500,000 if the drug successfully completes Phase II and III clinical trials and is approved for sale and marketing by the United States FDA. Future milestones on sales revenue are $1 million on the first $20 million in sales revenue, $2 million in the first-year cumulative sales revenue exceeds $100 million, $10 million in the first-year cumulative sales revenue exceeds $500 million, and $20 million in the first-year cumulative sales revenue exceeds $1 billion. We assessed whether the license should be capitalized and determined that the licensed program is in the early stage and therefore may not be recoverable; we expensed the license fee and will expense development costs until commercial viability is likely.
Johns Hopkins University – Prodrug License
In October 2022, we entered into an exclusive, worldwide, royalty-bearing license with JHU and the Institute of Organic Chemistry and Biochemistry (“IOCB”) of the Czech Academy of Sciences for rights to commercialize N-substituted prodrugs of mebendazole that demonstrate improved solubility and bioavailability. The license covers prodrug compositions and use for treating disease as claimed in multiple United States and worldwide patent applications. The term of the agreement began in October 2022 and continues until the date of expiration of the last to expire patent, or for 20 years from the effective date of the agreement if no patents are issued. The license can be terminated by us upon 90 days’ written notice, or by the licensor in the event of any material breach of the license that is not cured by the Company within 30 days.
In consideration for the rights granted to us under the license agreement, JHU and IOCB received a staggered upfront license fee of $100,000. We also reimbursed JHU and IOCB for previously incurred patent costs totaling $33,265 and will be responsible for reimbursing licensors for future patent costs. Under the terms of the license agreement, the licensors will be entitled to a four percent (4%) royalty on net sales subject to annual minimums upon first commercial sale of a licensed product, as well as sublicense or assignment fees in the event we sublicense or assign our rights to use the technology. The sublicense fee amount declines as we advance the clinical development of licensed technology. We are required to pay minimum annual royalties beginning in year 4 of the agreement. The minimum annual royalty for year 4 will be $5,000 (2026), increasing to $10,000 in year 5 (2027), $20,000 in year 6 (2028), $30,000 in year 7 (2029), and $50,000 in year 8 and subsequent years (2030 and beyond). We will be responsible for milestone payments for patent issuance of up to $50,000 and clinical development milestones up to and including approval of an NDA totaling up to $2.3 million. We will be required to pay a commercial milestone of $1 million once sales reach $20 million in the United States, $2 million when sales in the United States reach $100 million, $10 million when United States sales reach $500 million, and $20 million when United States sales exceed $1 billion. We assessed whether the license should be capitalized and determined that the licensed program is in the early stage and therefore may not be recoverable; we expensed the license fee and will expense development costs until commercial viability is likely.
| 17 |
In September 2023, the Company announced positive data in a preclinical study investigating the anti-cancer activity of a novel prodrug of mebendazole for the treatment of glioblastoma. The study assessed the relative efficacy of BF-222, a novel formulation of mebendazole that has been evaluated in clinical trials, and BF-223, a novel prodrug of mebendazole with improved solubility and bioavailability relative to BF-222, compared with placebo in mice that had been implanted with tumor cells as a model for human glioblastoma. Animals treated with BF-223 had an average survival time of 27.9 days compared with 27.3 days for mice treated with BF-222 and 23.4 days for mice given the placebo. Mice treated with BF-223 were administered 80% of the dose that mice treated with BF-222 received, and improved outcomes for both treatment groups were statistically significant compared to the placebo. In addition, animals treated with equivalent doses of BF-222 and BF-223 showed comparable and significant reduction in tumor growth compared to control animals during the study.
Competition
The pharmaceutical and biotechnology industries are characterized by rapidly advancing technologies, intense competition, and a strong emphasis on proprietary products. The immuno-oncology, neuroscience, and rare disease segments of the industry in particular are highly competitive. While we believe that our technology, development experience and scientific knowledge provide competitive advantages, we face potential competition from many different sources, including major pharmaceutical, specialty pharmaceutical, and biotechnology companies, academic institutions and governmental agencies, and public and private research institutions.
Many of our competitors may have significantly greater financial resources and expertise in research and development, manufacturing, preclinical studies, conducting clinical trials, obtaining regulatory approvals, and marketing approved medicines than we do. Mergers and acquisitions in the pharmaceutical, biotechnology, and diagnostic industries may result in even more resources being concentrated among a smaller number of our competitors. These competitors also compete with us in recruiting and retaining qualified scientific and management personnel and in establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to or necessary for our programs. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies.
The key competitive factors affecting the success of all of our product candidates, if approved, are likely to be their efficacy, safety, convenience, price, the effectiveness of companion diagnostics in guiding the use of related therapeutics, if any, the level of generic competition and the availability of reimbursement from government and other third-party payors.
Our commercial opportunity could be reduced or eliminated if our competitors develop and commercialize medicines that are safer, are more effective, have fewer or less severe side effects, are more convenient or are less expensive than any medicines we may develop. Our competitors also may obtain FDA or other regulatory approval for their medicines more rapidly than we may obtain approval for ours, which could result in our competitors establishing a strong market position before we are able to enter the market. In addition, our ability to compete may be affected in many cases by insurers or other third-party payors seeking to encourage the use of generic medicines. There are many generic medicines currently on the market for certain of the indications that we are pursuing, and additional generics are expected to become available over the coming years. If our therapeutic product candidates are approved, we expect that they will be priced at a significant premium over competitive generic medicines.
Any product candidates that we successfully develop and commercialize will compete with existing therapies and new therapies that may become available in the future. If the product candidates of our priority programs are approved for the indications for which we are currently planning clinical trials, they will compete with the drugs discussed below and will likely compete with other drugs currently in development.
| 18 |
bfLEAPTM
The analytics industry and application of AI/ML in healthcare is growing rapidly. Competition exists along the entire continuum of the drug development process from discovery to commercialization and beyond. We believe the weakness of the industry is the quality of the data and we believe bfLEAPTM provides several competitive advantages, that will position the Company for success, First, bfLEAPTM is highly scalable and can process data from small to extremely large, complex data sets without the need for additional code being developed. Second, it is adept at processing and analyzing incomplete data and making predictions that we do not believe other technologies are capable of doing. Third, bfLEAPTM has the ability to extract the most important features for analysis out of extremely large, complex data sets using unsupervised machine learning algorithms, thereby greatly simplifying complex problems. Since data quality is a problem that exists in the healthcare industry, we see these as major differentiators. The ability to make predictions and find relationships, patterns, and anomalies in extremely large, complex data sets has been demonstrated by JHU-APL in other applications and sectors. Finally, the algorithms used by bfLEAPTM are proprietary and protected, having been developed at JHU-APL. We believe most of the competitors rely on open-source algorithms and we also believe that we have already demonstrated our superiority via the August 2021 publication in DeepAI.org.
Government Regulation
The FDA does not currently require approval of AI/ML technologies used to aid in therapeutics, but that could change in the future. The FDA will regulate any clinical trials conducted by the Company.
Our clinical development programs will, in some cases, require regulatory review of preclinical and/or clinical data by the FDA or other governing agencies, and subsequent compliance with applicable federal, state, local, and foreign statutes and regulations. The results of the clinical trials that we conduct will be evaluated by the FDA and other regulatory bodies. The comments and approvals that are obtained are expected to lead to milestone payments under our agreements. Accordingly, our ability to navigate the regulatory process is extremely important to the success of the Company. We believe that we have a competitive advantage in this process due to primarily focusing on drug candidates that already have some level of success in clinical trials. Previous success of a particular candidate in trials combined with our precision medicine approach to clinical trial design using our bfLEAPTM platform, will de-risk the development process and improve the chances for success.
Government Regulation and Product Approval
Government authorities in the United States, at the federal, state and local level, and in other countries and jurisdictions extensively regulate, among other things, the research, development, testing, manufacture, quality control, approval, packaging, storage, recordkeeping, labeling, advertising, promotion, distribution, marketing, post-approval monitoring and reporting, and import and export of pharmaceutical products. The processes for obtaining regulatory approvals in the United States and in foreign countries and jurisdictions, along with subsequent compliance with applicable statutes and regulations and other regulatory authorities, require the expenditure of substantial time and financial resources.
FDA Approval Process
In the United States, pharmaceutical products are subject to extensive regulation by the FDA. The Federal Food, Drug, and Cosmetic Act (“FD&C Act”) and other federal and state statutes and regulations govern, among other things, the research, development, testing, manufacture, storage, recordkeeping, approval, labeling, promotion and marketing, distribution, post-approval monitoring and reporting, sampling and import and export of pharmaceutical products. Failure to comply with applicable United States requirements may subject a company to a variety of administrative or judicial sanctions, such as FDA refusal to approve pending new drug applications (“NDAs”), warning or untitled letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, civil penalties and criminal prosecution.
Pharmaceutical product development for a new product or certain changes to an approved product in the United States typically involves preclinical laboratory and animal tests, the submission to the FDA of an investigational new drug application (“IND”) which must become effective before clinical testing may commence, and adequate and well-controlled clinical trials to establish the safety and effectiveness of the drug for each indication for which FDA approval is sought. Satisfaction of FDA pre-market approval requirements typically takes many years and the actual time required may vary substantially based upon the type, complexity and novelty of the product or disease.
| 19 |
Preclinical tests include laboratory evaluation of product chemistry, formulation and toxicity, as well as animal trials to assess the characteristics and potential safety and efficacy of the product. The conduct of the preclinical tests must comply with federal regulations and requirements, including good laboratory practices. The results of preclinical testing are submitted to the FDA as part of an IND along with other information, including information about product chemistry, manufacturing and controls, and a proposed clinical trial protocol. Long-term preclinical tests, such as animal tests of reproductive toxicity and carcinogenicity, may continue after the IND is submitted. A 30-day waiting period after the submission of each IND is required prior to the commencement of clinical testing in humans. If the FDA has neither commented on nor questioned the IND within this 30-day period, the clinical trial proposed in the IND may begin. Clinical trials involve the administration of the investigational new drug to healthy volunteers or patients under the supervision of a qualified investigator. Clinical trials must be conducted: (i) in compliance with federal regulations; (ii) in compliance with good clinical practice, or GCP, an international standard meant to protect the rights and health of patients and to define the roles of clinical trial sponsors, administrators, and monitors; as well as (iii) under protocols detailing the objectives of the trial, the parameters to be used in monitoring safety and the effectiveness criteria to be evaluated. Each protocol involving testing on United States patients and subsequent protocol amendments must be submitted to the FDA as part of the IND.
Clinical trials to support NDAs for marketing approval are typically conducted in three sequential phases, but the phases may overlap. In Phase I, the initial introduction of the drug into healthy human subjects or patients, the drug is tested to assess metabolism, pharmacokinetics, pharmacological actions, side effects associated with increasing doses, and, if possible, early evidence of effectiveness. Phase II usually involves trials in a limited patient population to determine the effectiveness of the drug for a particular indication, dosage tolerance and optimum dosage, and to identify common adverse effects and safety risks. If a drug demonstrates evidence of effectiveness and an acceptable safety profile in Phase II evaluations, Phase III trials are undertaken to obtain the additional information about clinical efficacy and safety in a larger number of patients, typically at geographically dispersed clinical trial sites, to permit the FDA to evaluate the overall benefit-risk relationship of the drug and to provide adequate information for the labeling of the drug. In most cases, the FDA requires two adequate and well-controlled Phase III clinical trials to demonstrate the efficacy of the drug. A single Phase III trial with other confirmatory evidence may be sufficient in rare instances, such as where the study is a large multicenter trial demonstrating internal consistency and a statistically very persuasive finding of a clinically meaningful effect on mortality, irreversible morbidity, or prevention of a disease with a potentially serious outcome and confirmation of the result in a second trial would be practically or ethically impossible.
After completion of the required clinical testing, an NDA is prepared and submitted to the FDA. FDA approval of the NDA is required before marketing of the product may begin in the United States. The NDA must include the results of all preclinical, clinical and other testing and a compilation of data relating to the product’s pharmacology, chemistry, manufacture and controls. The cost of preparing and submitting an NDA is substantial. The submission of most NDAs is additionally subject to a substantial application user fee, and the applicant under an approved NDA is also subject to an annual program fee for each prescription product. These fees are typically increased annually. Sponsors of applications for drugs granted Orphan Drug Designation are exempt from these user fees.
The FDA may also refer applications for novel drug products, or drug products that present difficult questions of safety or efficacy, to an outside advisory committee, typically a panel that includes clinicians and other experts, for review, evaluation, and a recommendation as to whether the application should be approved. The FDA is not bound by the recommendation of an advisory committee, but it generally follows such recommendations.
Before approving an NDA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP. Additionally, the FDA will inspect the facility or the facilities at which the drug is manufactured. The FDA will not approve the product unless compliance with current good manufacturing practices (cGMPs) is satisfactory and the NDA contains data that provides substantial evidence that the drug is safe and effective in the indication studied.
Fast Track Designation
The FDA is required to facilitate the development, and expedite the review, of drugs that are intended for the treatment of a serious or life-threatening disease or condition for which there is no effective treatment and which demonstrates the potential to address unmet medical needs for the condition. Under the Fast Track program, the sponsor of a new drug candidate may request that the FDA designate the drug candidate for a specific indication as a Fast Track drug concurrent with, or after, the filing of the IND for the drug candidate. FDA must determine if the drug candidate qualifies for Fast Track Designation within 60 days of receipt of the sponsor’s request.
| 20 |
If a submission is granted Fast Track Designation, the sponsor may engage in more frequent interactions with the FDA, and the FDA may review sections of the NDA before the application is complete. This rolling review is available if the applicant provides, and the FDA approves, a schedule for the submission of the remaining information and the applicant pays applicable user fees. However, the FDA’s time period goal for reviewing an application does not begin until the last section of the NDA is submitted. While we may seek Fast Track Designation, there is no guarantee that we will be successful in obtaining any such designation. Even if we do obtain such designation, we may not experience a faster development process, review or approval compared to conventional FDA procedures. A Fast Track Designation does not ensure that the product candidate will receive marketing approval or that approval will be granted within any particular timeframe. Additionally, Fast Track Designation may be withdrawn by the FDA if the FDA believes that the designation is no longer supported by data emerging in the clinical trial process.
Post-Approval Requirements
Once an NDA is approved, a product will be subject to certain post-approval requirements. For instance, the FDA closely regulates the post-approval marketing and promotion of drugs, including standards and regulations for direct-to-consumer advertising, off-label promotion, industry-sponsored scientific and educational activities and promotional activities involving the internet. Drugs may be marketed only for the approved indications and in accordance with the provisions of the approved labeling.
Adverse event reporting and submission of periodic reports are required following FDA approval of an NDA. The FDA also may require post-marketing testing, known as Phase IV testing, REMS and surveillance to monitor the effects of an approved product, or the FDA may place conditions on an approval that could restrict the distribution or use of the product. In addition, quality control, drug manufacture, packaging and labeling procedures must continue to conform to cGMPs after approval. Drug manufacturers and certain of their subcontractors are required to register their establishments with FDA and certain state agencies. Registration with the FDA subjects entities to periodic unannounced inspections by the FDA, during which the Agency inspects manufacturing facilities to assess compliance with cGMPs. Accordingly, manufacturers must continue to expend time, money, and effort in the areas of production and quality-control to maintain compliance with cGMPs. Regulatory authorities may withdraw product approvals or request product recalls if a company fails to comply with regulatory standards, if it encounters problems following initial marketing, or if previously unrecognized problems are subsequently discovered.
Generic Competition
In seeking approval for a drug through an NDA, applicants are required to list with the FDA each patent whose claims cover the applicant’s product. Upon approval of a drug, each of the patents listed in the application for the drug is then published in the FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations, commonly known as the Orange Book. Drugs listed in the Orange Book can, in turn, be cited by potential generic competitors in support of approval of an abbreviated new drug application (“ANDA”). An ANDA provides for marketing of a drug product that has the same active ingredients in the same strengths and dosage form as the listed drug and has been shown through bioequivalence testing to be therapeutically equivalent to the listed drug. Other than the requirement for bioequivalence testing, ANDA applicants are not required to conduct, or submit results of, preclinical or clinical tests to prove the safety or effectiveness of their drug product. Drugs approved in this way are commonly referred to as “generic equivalents” to the listed drug and can often be substituted by pharmacists under prescriptions written for the original listed drug.
The ANDA applicant is required to certify to the FDA concerning any patents listed for the approved product in the FDA’s Orange Book. Specifically, the applicant must certify that (i) the required patent information has not been filed; (ii) the listed patent has expired; (iii) the listed patent has not expired but will expire on a particular date and approval is sought after patent expiration; or (iv) the listed patent is invalid or will not be infringed by the new product (a Paragraph IV certification). The ANDA applicant may also elect to submit a section viii statement certifying that its proposed ANDA label does not contain (or carve out) any language regarding the patented method-of-use rather than certify to a listed method-of-use patent. If the applicant does not challenge the listed patents or certifies that the listed patents will not be infringed by the new product, the ANDA application will not be approved until all the listed patents claiming the referenced product have expired. If the ANDA applicant has provided a Paragraph IV certification, the NDA and patent holders may then initiate a patent infringement lawsuit in response. The filing of a patent infringement lawsuit within 45 days of the receipt of such a certification automatically prevents the FDA from approving the ANDA until the earlier of 30 months, expiration of the patent, settlement of the lawsuit, or a decision in the infringement case that is favorable to the ANDA applicant.
| 21 |
Exclusivity
Upon NDA approval of a new chemical entity (“NCE”), that drug receives five years of marketing exclusivity during which the FDA cannot receive any ANDA seeking approval of a generic version of that drug. An ANDA may be submitted one year before NCE exclusivity expires if a Paragraph IV certification is filed. If there is no listed patent in the Orange Book, there may not be a Paragraph IV certification, and, thus, no ANDA may be filed before the expiration of the exclusivity period. Certain changes to a drug, such as the addition of a new indication to the package insert, can be the subject of a three-year period of exclusivity if the application contains reports of new clinical investigations (other than bioavailability studies) conducted or sponsored by the sponsor that were essential to approval of the application. The FDA cannot approve an ANDA for a generic drug that includes the change during the period of exclusivity.
Patent Term Extension
After NDA approval, owners of relevant drug patents may apply for up to a five-year patent extension. The allowable patent term extension is calculated as half of the drug’s testing phase (the time between IND application and NDA submission) and all of the review phase (the time between NDA submission and approval up to a maximum of five years). The time can be shortened if the FDA determines that the applicant did not pursue approval with due diligence. The total patent term after the extension may not exceed 14 years, and only one patent can be extended. For patents that might expire during the application phase, the patent owner may request an interim patent extension. An interim patent extension increases the patent term by one year and may be renewed up to four times. For each interim patent extension granted, the post-approval patent extension is reduced by one year. The director of the United States Patent and Trademark Office must determine that approval of the drug covered by the patent for which a patent extension is being sought is likely. Interim patent extensions are not available for a drug for which an NDA has not been submitted.
Other Healthcare Laws
In the United States, biotechnology company activities are subject to regulation by various federal, state and local authorities in addition to the FDA, including but not limited to, the Centers for Medicare & Medicaid Services (“CMS”), other divisions of the U.S. Department of Health and Human Services (e.g., the Office of Inspector General and the Office for Civil Rights), the U.S. Department of Justice (“DOJ”) and individual U.S. Attorney offices within the DOJ, and state and local governments. For example, research, sales, marketing, and scientific/educational grant programs have to comply with the anti-fraud and abuse provisions of the Social Security Act, the federal false claims laws, the privacy and security provisions of the Health Insurance Portability and Accountability Act (“HIPAA”) and similar state laws, each as amended, as applicable.
Also, many states have similar fraud and abuse statutes or regulations that apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor.
Data privacy and security regulations by both the federal government and the states in which business is conducted may also be applicable. HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, or HITECH, and its implementing regulations, imposes requirements relating to the privacy, security and transmission of individually identifiable health information. HIPAA requires covered entities to limit the use and disclosure of protected health information to specifically authorized situations and requires covered entities to implement security measures to protect health information that they maintain in electronic form. Among other things, HITECH made HIPAA’s security standards directly applicable to business associates, independent contractors or agents of covered entities that receive or obtain protected health information in connection with providing a service on behalf of a covered entity. HITECH also created four new tiers of civil monetary penalties, amended HIPAA to make civil and criminal penalties directly applicable to business associates, and gave state attorneys general new authority to file civil actions for damages or injunctions in federal courts to enforce the federal HIPAA laws and seek attorneys’ fees and costs associated with pursuing federal civil actions. In addition, state laws govern the privacy and security of health information in specified circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts.
| 22 |
Insurance Coverage and Reimbursement
Significant uncertainty exists as to the insurance coverage and reimbursement status of any products for which we may obtain regulatory approval. In the United States, sales of any product candidates for which regulatory approval for commercial sale is obtained will depend in part on the availability of coverage and adequate reimbursement from third-party payors. Third-party payors include government authorities and health programs in the United States such as Medicare and Medicaid, managed care providers, private health insurers and other organizations. These third-party payors are increasingly reducing reimbursements for medical products and services. The process for determining whether a payor will provide coverage for a drug product may be separate from the process for setting the reimbursement rate that the payor will pay for the drug product. Third-party payors may limit coverage to specific drug products on an approved list, or formulary, which might not include all of the FDA-approved drugs for a particular indication. A payor’s decision to provide coverage for a drug product does not imply that an adequate reimbursement rate will be approved. Further, coverage and reimbursement for drug products can differ significantly from payor to payor. As a result, the coverage determination process is often a time-consuming and costly process that will require us to provide scientific and clinical support for the use of our products to each payor separately, with no assurance that coverage and adequate reimbursement will be applied consistently or obtained in the first instance.
Human Capital Resources
As of December 31, 2025, the Company had 9 full-time employees, including its Chief Executive Officer, and 8 part-time employees, advisors, and consultants, including its Chief Financial Officer. None of these employees are covered by a collective bargaining agreement, and we believe our relationship with our employees is good. We also engage consultants on an as-needed basis to supplement existing staff.
We believe that our future success will depend, in part, on our continued ability to attract, hire and retain qualified personnel. In particular, we depend on the skills, experience and performance of our senior management and R&D personnel. We compete for qualified personnel with other healthcare technology, pharmaceutical, and healthcare companies, as well as universities and non-profit research institutions. We provide competitive compensation and benefits programs to help meet the needs of our employees. In addition to salaries, these programs include incentive compensation plans, healthcare and insurance benefits, a retirement plan, paid time off, and family leave, among others. We also use targeted equity-based grants with vesting conditions to facilitate retention of personnel, particularly for our key employees.
Properties
Currently, we do not own or lease any real property. All of our employees work virtually.
Legal Proceedings
We are not a party to any legal proceedings.
Corporate Information
BullFrog AI Holdings, Inc. was incorporated in the State of Nevada in February 2020. Our principal business address is 325 Ellington Blvd, Unit 317, Gaithersburg, MD 20878. Our website address is www.bullfrogai.com. The references to our website in this annual report are inactive textual references only. The information on our website is neither incorporated by reference into this annual report nor intended to be used in connection with this annual report. All our operations are currently conducted through BullFrog AI Holdings, Inc.
Available Information
We file annual, quarterly, and current reports, proxy statements and other information with the U.S. Securities Exchange Commission (the “SEC”). These filings are available to the public through the SEC’s website at http://www.sec.gov. All statements made in any of our securities filings, including all forward-looking statements or information, are made as of the date of the document in which the statement is included unless otherwise specified, and we do not assume or undertake any obligation to update any of those statements or documents unless we are required to do so by law.
| 23 |
ITEM 1A. RISK FACTORS
Investing in our securities involves a high degree of risk. You should carefully consider the risks and uncertainties described in the risk factors below, in addition to other information contained in or incorporated by reference into this Annual Report. Our business, financial condition and results of operations could be materially adversely affected by the materialization of any of these risks. Additional risks not currently known to us or that we currently deem to be immaterial also may materially adversely affect our business, financial condition and results of operations in future periods.
Risks Related to Liquidity, the Company’s Business and Industry
We have a limited operating history upon which you can evaluate our performance, and accordingly, our prospects must be considered in light of the risks that any new company encounters.
We were incorporated under the laws of Nevada in February 2020. Accordingly, we have no significant history upon which an evaluation of our prospects and future performance can be made. Our proposed operations are subject to all of the business risks associated with a new enterprise. The likelihood of our creation of a viable business must be considered in light of the problems, expenses, difficulties, complications, and delays frequently encountered in connection with the inception of a business, operation in a competitive industry, and the continued development of our technology and the results of our clinical data. We anticipate that our operating expenses will increase in the near future. There can be no assurances that we will ever operate profitably. You should consider our business, operations and prospects in light of the risks, expenses and challenges faced as an early-stage company.
Our independent registered public accounting firm has included an explanatory paragraph relating to our ability to continue as a going concern in its report on our audited financial statements included in this annual report.
The report from our independent registered public accounting firm for the year ended December 31, 2025, includes an explanatory paragraph stating that we have incurred significant losses and need to raise additional funds to meet our obligations and sustain our operations. These conditions raise substantial doubt about our ability to continue as a going concern. As of December 31, 2025, we had approximately $2.3 million in cash, which includes restricted cash of $0.1 million held by a financial institution as collateral for the Company’s corporate credit card program, and an accumulated deficit of approximately $23.3 million. We believe that our existing cash and cash equivalents as of December 31, 2025 will not be sufficient to fund our planned operations for at least a year beyond the filing date of the consolidated financial statements. Our recurring losses from operations since inception and required additional funding to finance our operations raise substantial doubt about our ability to continue as a going concern. These conditions could materially limit our ability to raise additional funds through the issuance of new debt or equity securities or otherwise. There is no assurance that sufficient financing will be available when needed, or at all, to allow us to continue as a going concern. The perception that we may not be able to continue as a going concern may also make it more difficult to operate our business due to concerns about our ability to meet our contractual obligations. Our ability to continue as a going concern is contingent upon, among other factors, the sale of our securities. There is no assurance that sufficient financing will be available when needed, or at all, to allow us to continue as a going concern.
If we are unable to secure additional capital, we may be required to curtail our clinical and research and development initiatives and take additional measures to reduce costs in order to conserve our cash in amounts sufficient to sustain operations and meet our obligations. These measures could cause significant delays in our clinical and regulatory efforts, which is critical to the realization of our business plan. The consolidated financial statements do not include any adjustments that may be necessary should we be unable to continue as a going concern. It is not possible for us to predict at this time the potential success of our business. The revenue and income potential of our proposed business and operations are currently unknown. If we cannot continue as a viable entity, you may lose some or all of your investment.
Future sales or issuances of our common stock in the public markets, or the perception of such sales, could depress the trading price of our common stock.
The sale of a substantial number of shares of our common stock or other securities convertible into or exchangeable for our common stock in the public markets, or the perception that such sales could occur, could depress the market price of our common stock and impair our ability to raise capital through the sale of additional equity securities. We may sell large quantities of our common stock at any time in one or more separate offerings. We cannot predict the effect that future sales of common stock or other equity-related securities would have on the market price of our common stock.
We are currently listed on the Nasdaq Capital Market. If we are unable to maintain listing of our securities on Nasdaq or any stock exchange, our stock price could be adversely affected and the liquidity of our stock and our ability to obtain financing could be impaired and it may be more difficult for our shareholders to sell their securities.
Although our common stock is currently listed on the Nasdaq Capital Market, we may not be able to continue to meet the exchange’s minimum listing requirements or those of any other national exchange. The listing rules of Nasdaq require listing issuers to comply with certain standards in order to remain listed on its exchange. For instance, on August 21, 2025, we received a letter from the listing staff of The Nasdaq Stock Market LLC (“Nasdaq”) that we were no longer in compliance with the minimum stockholders’ equity requirement for continued listing on Nasdaq pursuant to Nasdaq Listing Rule 5550(b)(1) (the “Stockholders’ Equity Rule”). The Stockholders’ Equity Rule requires companies listed on the Nasdaq Capital Market to maintain stockholders’ equity of at least $2,500,000 or to meet alternatives of market value of listed securities or net income from continuing operations, which we do not currently meet. In response to our proposed compliance plan, on October 7, 2025, we received a letter (the “Extension Letter”) from Nasdaq informing us that, based on the plan, Nasdaq had granted our request for an extension until February 17, 2026, to comply with the Stockholders’ Equity Requirement. On February 19, 2026, we received a further notice from Nasdaq notifying us that Nasdaq determined that we had not met the terms of the extension. We thereafter timely requested a hearing before an independent Nasdaq Hearings Panel that automatically stayed any suspension or delisting action pending the hearing and the expiration of any extension period granted by the Panel following the hearing. At the hearing, we plan to present additional details of our compliance plan and provide an update on our efforts to regain compliance. We will also request additional time to complete the steps of our compliance plan and regain compliance with all applicable Nasdaq Listing Rules.
| 24 |
In addition, on February 10, 2026, we received a letter from Nasdaq notifying us that, for the last 30 consecutive business days, the closing bid price for our common stock, par value $0.00001 per share, was below $1.00 per share, which is the minimum closing bid price required for continued listing on the Nasdaq Global Market (the “Minimum Bid Price Requirement”) pursuant to Nasdaq Listing Rule 5550(a)(2) (the “Bid Price Notice”). In accordance with Nasdaq Listing Rule 5810(c)(3)(A), we were provided a compliance period of 180 calendar days from the date of the Bid Price Notice, or until August 10, 2026, to regain compliance with the Minimum Bid Price Requirement. If we do not regain compliance during the initial 180-calendar day compliance period, we may be provided a second 180-calendar day period to regain compliance. If we do not regain compliance within the allotted compliance periods, including any extensions that may be granted by Nasdaq, our listed securities will be subject to delisting.
There can be no assurance that the Nasdaq Hearings Panel will grant our request for additional time to regain compliance with Nasdaq listing rules or that, if the Panel does grant our request, we will be able to regain compliance with the applicable Nasdaq listing requirements. If, for any reason, we are unable to regain compliance with Nasdaq’s listing standards, our securities would be subject to delisting by Nasdaq. In such case, unless we are able to list on another national securities exchange, a reduction in some or all of the following may occur, each of which could have a material adverse effect on our shareholders:
| ● | the liquidity of our common stock; | |
| ● | the market price of our common stock; | |
| ● | our ability to obtain financing for the continuation of our operations; | |
| ● | the number of investors that could consider investing in our common stock; | |
| ● | the number of market makers in our common stock; | |
| ● | the availability of information concerning the trading prices and volume of our common stock; and | |
| ● | the number of broker-dealers willing to execute trades in shares of our common stock. |
If we are unable to attract and retain key management, scientific personnel and advisors, we may not achieve our business objectives.
Our success depends on the availability and contributions of members of our senior management team. The loss of services of any of these individuals could delay, reduce or prevent our drug development and other business objectives. Furthermore, recruiting and retaining qualified scientific personnel to perform drug development work will be critical to our success. We face intense competition for qualified individuals from numerous pharmaceutical and biotechnology companies, universities, governmental entities and other public and private research institutions. We may be unable to attract and retain these individuals, and our failure to do so could materially adversely affect our business and financial condition.
The development of our technology, products, and services is highly competitive.
We face competition with respect to any products that we may seek to develop or commercialize in the future. Our competitors include major companies worldwide. Many of our competitors have significantly greater financial, technical and human resources than we have and superior expertise in research and development and marketing approved products and services and thus may be better equipped than us to develop and commercialize products and services. These competitors also compete with us in recruiting and retaining qualified personnel and acquiring technologies. Smaller or early stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. Accordingly, our competitors may commercialize products more rapidly or effectively than we are able to, which would adversely affect our competitive position, the likelihood that our products and services will achieve initial market acceptance and our ability to generate meaningful additional revenues from our products.
From time to time, third parties may claim that one or more of our products or services infringe their intellectual property rights.
Any dispute or litigation regarding patents or other intellectual property could be costly and time consuming due to the uncertainty of intellectual property litigation and could divert our management and key personnel from our business operations. A claim of intellectual property infringement could force us to enter into a costly or restrictive license agreement, which might not be available under acceptable terms or at all, could require us to redesign our products, which would be costly and time-consuming, and/or could subject us to an injunction against development and sale of certain of our products or services. We may have to pay substantial damages, including damages for past infringement if it is ultimately determined that our products or services infringe on a third party’s proprietary rights. Even if these claims are without merit, defending a lawsuit takes significant time, may be expensive and may divert management’s attention from other business concerns. Any public announcements related to litigation or interference proceedings initiated or threatened against us could cause our business to be harmed. Our intellectual property portfolio may not be useful in asserting a counterclaim, or negotiating a license, in response to a claim of intellectual property infringement. In certain of our businesses we rely on third party intellectual property licenses and we cannot ensure that these licenses will be available to us in the future on favorable terms or at all.
Although dependent on certain key personnel, we do not have any key man life insurance policies on any such people.
We are dependent on Vininder Singh, our Chief Executive Officer, in order to conduct our operations and execute our business plan and the loss of Vininder Singh or any member of the board of directors or executive officer could harm our business, financial condition, cash flow and results of operations; however, we have not purchased any insurance policies with respect to those individuals in the event of their death or disability. Therefore, if Vininder Singh or any member of the board of directors or an executive officer dies or become disabled, we will not receive any compensation to assist with such person’s absence. The loss of such person could negatively affect us and our operations.
| 25 |
New product development involves a lengthy, expensive and complex process.
We may be unable to develop or commercialize any product candidates. Moreover, even if we develop such candidates, they may be subject to significant regulatory review, approval and other government regulations. There can be no assurance that our technologies will be capable of developing and commercializing products at all. New product development involves a lengthy, expensive and complex process and we currently have no fully validated diagnostic candidates. In addition, before we can commercialize any new product candidates, we will need to:
| ● | conduct substantial research and development; | |
| ● | conduct validation studies; | |
| ● | expend significant funds; | |
| ● | develop and scale-up our laboratory processes; and | |
| ● | obtain regulatory approval and acceptance of our product candidates. |
This process involves a high degree of risk and takes several years. Our product development efforts may fail for many reasons, including:
| ● | failure of the product or service at the research or development stage; and | |
| ● | lack of clinical validation data to support the effectiveness of the product or service. |
Few research and development projects result in commercial products, and perceived viability in early clinical trials often is not replicated in later studies. At any point, we may abandon development of a product candidate or we may be required to expend considerable resources repeating clinical trials, which would adversely impact the timing for generating potential revenues from those product candidates. In addition, as we develop product candidates, we will have to make significant investments in product development, marketing and sales resources.
We may not be able to conduct clinical trials necessary to increase the value of our proposed products and formulations.
In order to conduct clinical trials that are necessary to obtain approval of a product by the FDA, it is necessary to receive clearance from the FDA to conduct such clinical trials. The FDA can halt clinical trials at any time for safety reasons or because we or our clinical investigators do not follow the FDA’s requirements for conducting clinical trials. If we are unable to receive clearance to conduct clinical trials or the trials are halted by the FDA, the likelihood of our ability to sell or license certain of our products would be greatly reduced as it is the FDA approval which will enhance the value of our products.
Our ability to resell and/or license certain of our products will depend upon successful clinical trials.
Only a small number of research and development programs result in the development of a product that obtains FDA approval. Success in preclinical work or early stage clinical trials does not ensure that later stage or larger scale clinical trials will be successful. Conducting clinical trials is a complex, time-consuming and expensive process. Our ability to complete our clinical trials in a timely fashion depends, in large part, on a number of key factors including protocol design, regulatory and institutional review board approval, the rate of patient enrollment in clinical trials, and compliance with extensive current Good Clinical Practices. If we fail to adequately manage the design, execution and regulatory aspects of our clinical trials, our studies and ultimately our regulatory approvals may be delayed, or we may fail to gain approval for our product candidates. Clinical trials may indicate that our product candidates have harmful side effects or raise other safety concerns that may significantly reduce the likelihood of regulatory approval, result in significant restrictions on use and safety warnings in any approved label, adversely affect placement within the treatment paradigm, or otherwise significantly diminish the commercial potential of the product candidate. Also, positive results in a registrational trial may not be replicated in any subsequent confirmatory trials. Even if later stage clinical trials are successful, regulatory authorities may disagree with our view of the data or require additional studies, and may fail to approve or delay approval of our product candidates or may grant marketing approval that is more restricted than anticipated, including indications for a narrower patient population than expected and the imposition of safety monitoring or educational requirements or risk evaluation and mitigation strategies. In addition, if another Company is the first to file for marketing approval of a competing drug candidate, that Company may ultimately receive marketing exclusivity for its drug candidate, thereby reducing the value of our product.
| 26 |
We face significant competition from other biotechnology and pharmaceutical companies.
While we believe that our technology, development experience and scientific knowledge provide competitive advantages, we face potential competition from many different sources, including major pharmaceutical, specialty pharmaceutical, and biotechnology companies, academic institutions and governmental agencies, and public and private research institutions. Many of our existing or potential competitors have substantially greater financial, technical and human resources than we do and significantly greater experience in the development of drug candidates as well as in obtaining regulatory approvals of those drug candidates in the United States and in foreign countries.
Mergers and acquisitions in the pharmaceutical and biotechnology industries could result in even more resources being concentrated among a small number of our competitors. Competition may increase further as a result of advances in the commercial applicability of technologies and greater availability of capital for investment in these industries. Our competitors may succeed in developing, acquiring or licensing, on an exclusive basis, drug candidates that are more effective or less costly than any drug candidate that we may develop.
Our ability to compete successfully will depend largely on our ability to:
| ● | identify drugs that have suffered set backs in the clinical development and regulatory process which we believe can be assisted by our platform’s ability to design a better study group; | |
| ● | attract qualified scientific, product development and commercial personnel; | |
| ● | obtain patent or other proprietary protection for our drugs and technologies; | |
| ● | obtain required regulatory approvals; | |
| ● | successfully collaborate with pharmaceutical companies in the discovery, development and commercialization of new drugs; and | |
| ● | negotiate competitive pricing and reimbursement with third party payors. |
The availability of our competitors’ technologies could limit the demand and the price we are able to charge for our services and for any drug candidate we develop. The inability to compete with existing or subsequently introduced drug development technologies would have a material adverse impact on our business, financial condition and prospects.
Established pharmaceutical companies and research institutions may invest heavily to accelerate discovery and development of novel compounds or to in license novel compounds that could make bfLEAP™ less competitive, which would have a material adverse impact on our business.
We may not be able to acquire the rights to any failed drugs or we may not be able to rescue failed drugs through analysis due to our technology or the lack of clinical data.
Our business model is based on the use of AI/ML technology, which technology may not uncover actionable insights or we may not be able to access sufficient clinical data to uncover such insights that lead to a successful project, clinical trial, or product. The failure of such projects, clinical trials or products would result in a loss of revenue from one of our three sources, which could have a material adverse impact on our business as a whole.
We may not succeed in acquiring the rights to failed drugs, which could limit one of our main sources of revenue.
Our business model is partly based on our ability to acquire drugs that have failed to pass Phase II or Phase III of the FDA approval process; however, there is no guarantee that we will be able to acquire the rights to such drugs, which would significantly impact our ability to generate revenue and, as a result, would have a material adverse impact on our business.
We intend to invest in early stage experimental technologies which have a high risk of failure.
To continue supporting our business model, we intend to invest in early stage and experimental technologies, some or all of which may not be useful to us. There is a risk that we will invest in technology that will not ultimately contribute to the success of our projects, which could have a material adverse impact on our business.
We are dependent on our collaborative agreements for the development of products and business development, which exposes us to the risk of reliance on the viability of third parties.
In conducting our research and development activities, we currently rely, and will in the future rely, on collaborative agreements with third parties such as manufacturers, contract research organizations, commercial partners, universities, governmental agencies and not-for-profit organizations for both strategic and financial resources. The loss of, or failure to perform by us or our partners under, any applicable agreements or arrangements, or our failure to secure additional agreements for other products in development, would substantially disrupt or delay our research and development and commercialization activities. Any such loss would likely increase our expenses and materially harm our business, financial condition and results of operations.
| 27 |
We will need to outsource any clinical trial activities as we usually perform only a small portion of the start-up activities in-house.
We rely on independent third-party contract research organizations (CROs) to perform our clinical studies, including document preparation, site identification, screening and preparation, pre-study visits, training, program management and bioanalytical analysis. Many important aspects of the services performed for us by the CROs are out of our direct control. If there is any dispute or disruption in our relationship with our CROs, our clinical trials could suffer or be delayed.
We are a biotechnology company with no significant revenue. We have incurred operating losses since our inception, and we expect to incur losses for the foreseeable future and may never achieve profitability.
We have incurred significant operating losses since our inception. To date, we have not generated significant revenue and we may not generate significant revenue from sales of our clinical analytics services or drug candidates for the foreseeable future. We expect to continue to incur significant operating losses, and we anticipate that our losses may increase substantially as we expand our drug development programs.
To achieve profitability, we must enter into collaborations with companies that are developing drugs and/or successfully develop and obtain regulatory approval for one or more drugs and effectively commercialize any drugs we develop. Even if we succeed in entering into collaborations and/or developing and commercializing one or more drug candidates, we may not be able to generate sufficient revenue and we may never be able to achieve or sustain profitability.
We will continue to require additional capital for the foreseeable future. If we are unable to raise additional capital when needed, we may be forced to delay, reduce or eliminate our drug acquisition efforts.
We expect to continue to incur significant operating expenses in connection with our ongoing activities, including conducting clinical trials and seeking regulatory approval of drug candidates. Our ongoing future capital requirements will depend on numerous factors, including:
| ● | the rate of progress, results and costs of completion of clinical trials of drug candidates; | |
| ● | the size, scope, rate of progress, results and costs of completion of any potential future clinical trials and preclinical tests of our drug candidates that we may initiate; | |
| ● | the costs of obtaining regulatory approval of drug candidates; | |
| ● | the scope, prioritization and number of drug development programs we pursue; | |
| ● | the costs for preparing, filing, prosecuting, maintaining and enforcing our intellectual property rights and defending intellectual property-related claims; | |
| ● | the extent to which we acquire or in-license other products and technologies and the costs to be able to obtain regulatory approval of such products; | |
| ● | our ability to establish strategic collaborations and licensing or other arrangements on terms favorable to us; and | |
| ● | competing technological and market developments. |
Any additional fundraising efforts may divert our management from their day to day activities, which may adversely affect our ability to identify and acquire new drug candidates and to further the regulatory process of such products. Our ability to raise additional funds will depend, in part, on the success of our product development activities and other factors related to financial, economic and market conditions, many of which are beyond our control. There can be no assurance that we will be able to raise additional capital when needed or on terms that are favorable to us, if at all. If adequate funds are not available on a timely basis, we may be forced to:
| ● | delay, reduce the scope of or eliminate one or more of our drug development programs; | |
| ● | limit the number of new products that we acquire or relinquish, license or otherwise dispose of rights on terms that are less favorable than if we were able to further the regulatory approval process; or | |
| ● | liquidate and dissolve the Company. |
If our operating plans change, we may require additional capital sooner than planned. Such additional financing may not be available when needed or on terms favorable to us. In addition, we may seek additional capital due to favorable market conditions or strategic considerations, even if we believe we have sufficient funds for our current and future operating plan.
We face significant risks related to the availability, cost, and performance of the computational infrastructure required to train and deploy AI models.
Training and operating large-scale AI models requires access to substantial and specialized computational resources, including high-performance graphics processing units, custom accelerators, and large-scale data center capacity. The global supply of these resources is constrained, and we depend on a limited number of suppliers for critical hardware components. Any disruption to the supply chain for AI-specialized chips, whether due to geopolitical tensions, export controls, manufacturing constraints, natural disasters, or supplier-specific issues, could materially impair our ability to train new models, scale our services, or meet customer demand. The cost of compute has risen substantially and may continue to increase as competition for scarce resources intensifies. We also rely on cloud infrastructure providers for a significant portion of our computing needs, and any disruption to these services, adverse changes to their pricing or terms, or their decision to prioritize their own competing AI offerings could adversely affect our operations. The capital expenditure required to build or secure proprietary compute infrastructure is substantial, and there can be no assurance that our investments in such infrastructure will yield adequate returns.
| 28 |
Export controls, trade restrictions, and national security regulations may limit our ability to operate in certain markets and access critical technologies.
Our business is subject to export controls and trade restrictions imposed by the United States and other governments that may limit our ability to deploy AI products and services in certain jurisdictions, collaborate with foreign researchers, or access critical technologies and components. The U.S. government has imposed and may further expand export controls on advanced AI chips, semiconductor manufacturing equipment, and AI model weights, particularly with respect to China and other countries of concern. These restrictions are evolving rapidly and may be expanded to cover additional technologies, end users, or jurisdictions. Compliance with export controls across multiple jurisdictions is complex and resource-intensive, and violations could result in significant civil and criminal penalties, loss of export privileges, and reputational harm. Retaliatory trade measures by foreign governments could also restrict our market access or supply chains. Additionally, emerging national security reviews of AI technologies, including reviews by the Committee on Foreign Investment in the United States, may impose restrictions on our ability to accept foreign investment, form partnerships, or serve certain customers.
The capital requirements for AI development are substantial and increasing, and we may be unable to secure adequate financing on acceptable terms.
Developing, training, and deploying state-of-the-art AI models requires a large and growing capital investment. Training runs for frontier AI models can cost tens to hundreds of millions of dollars in compute alone, and these costs are expected to increase as models grow in scale and complexity. In addition to training costs, we must invest heavily in inference infrastructure, data acquisition, talent compensation, and research and development to remain competitive. Our future capital needs will depend on many factors, including the pace of technological change, competitive dynamics, customer growth, and the regulatory environment. We may need to raise additional capital through equity offerings, debt financing, or strategic partnerships, and there can be no assurance that such financing will be available on acceptable terms or at all. Market conditions, investor sentiment toward AI companies, and our financial performance could all adversely affect our ability to raise capital. If adequate financing is not available, we may be forced to delay or scale back our research and development efforts, reduce our infrastructure investments, or otherwise limit our growth, any of which could materially and adversely affect our competitive position and long-term prospects.
We may be subject to laws and regulations governing our use of artificial intelligence.
The use of AI in health care, and particularly the drug development process, continues to increase and evolve. While there currently is no Federal law governing the use of AI in health care or otherwise, several states and Federal agencies use existing regulations to govern the use of AI and enforce related privacy violations, and it is possible that governing legislation and regulations may be forthcoming given that President Trump has issued multiple AI-related Executive Orders, including an AI Action Plan on July 23, 2025 through Executive Order, “Promoting the Export of the American AI Technology Stock,” and a December 11, 2025 Executive Order, “Ensuring a National Policy Framework for Artificial Intelligence.” The FDA has indicated its intention to regulate the use of AI by drug manufacturers through multiple announcements, including its January 2025 draft guidance “Considerations for the Use of Artificial Intelligence to Support Regulatory Decision-Making for Drug and Biological Products,” and its January 2026 “Guiding Principles of Good AI Practice in Drug Development,” which establishes ten high-level guiding principles concerning future use and regulation of AI by pharmaceutical manufacturers.
As with privacy and security laws, we cannot predict the ultimate result of proposals to govern and regulate AI and any related enforcement actions, or the potential costs any compliance obligations may have on us. Violation of any applicable AI-related laws or regulations could have a material adverse effect on our business, financial condition and operating results.
Unfavorable global economic conditions could adversely affect our business, financial condition or results of operations.
Our results of operations could be adversely affected by general conditions in the global economy and in the global financial markets. Portions of our future clinical trials may be conducted outside of the United States and unfavorable economic conditions resulting in the weakening of the U.S. dollar would make those clinical trials costlier to operate. Furthermore, the most recent global financial crisis caused extreme volatility and disruptions in the capital and credit markets. A severe or prolonged economic downturn, due to factors including the effects of health epidemics and pandemics, such as COVID-19, geopolitical events, such as recent U.S. and Israeli military operations involving Iran and related disruption in the region, the Russian invasion of Ukraine, conflict in the Middle East and any other related global escalation of geopolitical tensions, inflationary pressures, interest rate volatility and domestic or international trade policy could result in a variety of risks to our business, including a reduced ability to raise additional capital when needed on acceptable terms, if at all. A weak or declining economy or international trade disputes could also strain our suppliers, some of which are located outside of the United States, possibly resulting in supply disruption. Any of the foregoing could harm our business and we cannot anticipate all of the ways in which the current economic climate and financial market conditions could adversely impact our business.
| 29 |
We are increasingly dependent on information technology systems to operate our business and a cyberattack or other breach of our systems, or those of third parties on whom we may rely, could subject us to liability or interrupt the operation of our business.
We are increasingly dependent on information technology systems to operate our business. A breakdown, invasion, corruption, destruction or interruption of critical information technology systems by employees, others with authorized access to our systems or unauthorized persons could negatively impact operations. In the ordinary course of business, we collect, store and transmit confidential information and it is critical that we do so in a secure manner to maintain the confidentiality and integrity of such information. Additionally, we outsource certain elements of our information technology systems to third parties. As a result of this outsourcing, our third party vendors may or could have access to our confidential information making such systems vulnerable. Data breaches of our information technology systems, or those of our third party vendors, may pose a risk that sensitive data may be exposed to unauthorized persons or to the public. For example, the loss of clinical trial data from completed or ongoing clinical trials or preclinical studies could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. While we believe that we have taken appropriate security measures to protect our data and information technology systems, and have been informed by our third party vendors that they have as well, there can be no assurance that our efforts will prevent breakdowns or breaches in our systems, or those of our third party vendors, that could materially adversely affect our business and financial condition.
We face risks associated with security breaches or cyberattacks.
We face risks associated with security breaches or cyber-attacks of our computer systems or those of our third-party representatives, vendors, and service providers. Armed conflicts in the Middle East and between Russia and Ukraine, and tensions with countries such as Iran and North Korea and resulting geopolitical uncertainties also could result in an increase in cyberattacks that could either directly or indirectly impact our operations, such as the recent cyberattacks for which Iran has taken credit for against U.S. medical device manufacturer Stryker. Although we have implemented security procedures and controls to address these threats, such as firewalls, encryption, access controls, and employee training programs, cybersecurity threats are dynamic and evolving and our systems may still be vulnerable to theft, loss or misuse of data, including proprietary or confidential information, relating to our business, products, employees, suppliers and customers; disruption due to computer viruses and programming errors; attacks by third parties including destruction of data or demanding ransom to return control of our systems and services; or similar disruptive problems.
Risks Related to Intellectual Property Rights
We rely on various intellectual property rights, including patents and licenses in order to operate our business.
Our intellectual property rights may not be sufficiently broad or otherwise may not provide us a significant competitive advantage. In addition, the steps that we have taken to maintain and protect our intellectual property may not prevent it from being challenged, invalidated, circumvented or designed-around, particularly in countries where intellectual property rights are not highly developed or protected. In some circumstances, enforcement may not be available to us because an infringer has a dominant intellectual property position or for other business reasons, or countries may require compulsory licensing of our intellectual property. Our failure to obtain or maintain intellectual property rights that convey competitive advantage, adequately protect our intellectual property or detect or prevent circumvention or unauthorized use of such property, could adversely impact our competitive position and results of operations. We also rely on nondisclosure and noncompetition agreements with employees, consultants and other parties to protect, in part, trade secrets and other proprietary rights. There can be no assurance that these agreements will adequately protect our trade secrets and other proprietary rights and will not be breached, that we will have adequate remedies for any breach, that others will not independently develop substantially equivalent proprietary information or that third parties will not otherwise gain access to our trade secrets or other proprietary rights.
As we expand our business, protecting our intellectual property will become increasingly important. The protective steps we have taken may be inadequate to deter our competitors from using our proprietary information. In order to protect or enforce our patent rights, we may be required to initiate litigation against third parties, such as infringement lawsuits. Also, these third parties may assert claims against us with or without provocation. These lawsuits could be expensive, take significant time and could divert management’s attention from other business concerns. The law relating to the scope and validity of claims in the technology field in which we operate is still evolving and, consequently, intellectual property positions in our industry are generally uncertain. We cannot assure you that we will prevail in any of these potential suits or that the damages or other remedies awarded, if any, would be commercially valuable.
| 30 |
We could be negatively impacted if found to have infringed on intellectual property rights.
Technology companies, including many of our competitors, frequently enter into litigation based on allegations of patent infringement or other violations of intellectual property rights. In addition, patent holding companies seek to monetize patents they have purchased or otherwise obtained. As we grow, the intellectual property rights claims against us will likely increase. We intend to vigorously defend infringement actions in court and before the U.S. International Trade Commission. The plaintiffs in these actions frequently seek injunctions and substantial damages. Regardless of the scope or validity of such patents or other intellectual property rights, or the merits of any claims by potential or actual litigants, we may have to engage in protracted litigation. If we are found to infringe on one or more patents or other intellectual property rights, regardless of whether we can develop non-infringing technology, we may be required to pay substantial damages or royalties to a third-party, or we may be subject to a temporary or permanent injunction prohibiting us from marketing or selling certain products. In certain cases, we may consider the desirability of entering into licensing agreements, although no assurance can be given that such licenses can be obtained on acceptable terms or that litigation will not occur. These licenses may also significantly increase our operating expenses. Regardless of the merit of particular claims, litigation may be expensive, time-consuming, disruptive to our operations and distracting to management. In recognition of these considerations, we may enter into arrangements to settle litigation. If one or more legal matters were resolved against us, our consolidated financial statements for that reporting period could be materially adversely affected. Further, such an outcome could result in significant compensatory, punitive or trebled monetary damages, disgorgement of revenue or profits, remedial corporate measures or injunctive relief against us that could adversely affect our financial condition and results of operations.
We rely heavily on our technology and intellectual property, but we may be unable to adequately or cost-effectively protect or enforce our intellectual property rights, thereby weakening our competitive position and increasing operating costs.
To protect our rights in our services and technology, we rely on a combination of copyright and trademark laws, patents, trade secrets, confidentiality agreements and protective contractual provisions. We also rely on laws pertaining to trademarks and domain names to protect the value of our corporate brands and reputation. Despite our efforts to protect our proprietary rights, unauthorized parties may copy aspects of our services or technology, obtain and use information, marks, or technology that we regard as proprietary, or otherwise violate or infringe our intellectual property rights. In addition, it is possible that others could independently develop substantially equivalent intellectual property. If we do not effectively protect our intellectual property, or if others independently develop substantially equivalent intellectual property, our competitive position could be weakened.
Effectively policing the unauthorized use of our services and technology is time-consuming and costly, and the steps taken by us may not prevent misappropriation of our technology or other proprietary assets. The efforts we have taken to protect our proprietary rights may not be sufficient or effective, and unauthorized parties may copy aspects of our services, use similar marks or domain names, or obtain and use information, marks, or technology that we regard as proprietary. We may have to litigate to enforce our intellectual property rights, to protect our trade secrets, or to determine the validity and scope of others’ proprietary rights, which are sometimes not clear or may change. Litigation can be time consuming and expensive, and the outcome can be difficult to predict.
We rely on agreements with third parties to provide certain services, goods, technology, and intellectual property rights necessary to enable us to implement some of our applications.
Our ability to implement and provide our applications and services to our clients depends, in part, on services, goods, technology, and intellectual property rights owned or controlled by third parties. These third parties may become unable to or refuse to continue to provide these services, goods, technology, or intellectual property rights on commercially reasonable terms consistent with our business practices, or otherwise discontinue a service important for us to continue to operate our applications. If we fail to replace these services, goods, technologies, or intellectual property rights in a timely manner or on commercially reasonable terms, our operating results and financial condition could be harmed. In addition, we exercise limited control over our third-party vendors, which increases our vulnerability to problems with technology and services those vendors provide. If the services, technology, or intellectual property of third parties were to fail to perform as expected, it could subject us to potential liability, adversely affect our renewal rates, and have an adverse effect on our financial condition and results of operations.
| 31 |
If any third-party owners of intellectual property we may license in the future do not properly maintain or enforce the patents underlying such licenses, our competitive position and business prospects will be harmed.
We may enter into licenses for third-party intellectual property in the future. Our success will depend in part on the ability of our licensors to obtain, maintain and enforce patent protection for their intellectual property, in particular, those patents to which we have secured exclusive rights.
If applicable, our licensors may not successfully prosecute the patent applications to which we are licensed. Even if patents issue in respect of any such patent applications, our licensors may fail to maintain these patents, may determine not to pursue litigation against other companies that are infringing these patents, or may pursue such litigation less aggressively than we would. In addition, our licensors may terminate their agreements with us in the event we breach the applicable license agreement and fail to cure the breach within a specified period of time. Without protection for the intellectual property we license, other companies might be able to offer substantially identical products for sale, which could materially adversely affect our competitive business position, business prospects and financial condition.
Because our research and development of drug candidates often incorporates compounds and other information that is the intellectual property of third parties, we depend on continued access to such intellectual property to conduct and complete our preclinical and clinical research and commercialize the drug candidates that result from this research. We expect that future licenses would impose numerous obligations on us. For example, under our existing and future license agreements, we may be required to pay (i) annual maintenance fees until a drug candidate is sold for the first time, (ii) running royalties on net sales of drug candidates, (iii) minimum annual royalties after a drug candidate is sold for the first time, and (iv) one-time payments upon the achievement of specified milestones. We may also be required to reimburse patent costs incurred by the licensor, or we may be obligated to pay additional royalties, at specified rates, based on net sales of our drug candidates that incorporate the licensed intellectual property rights. We may also be obligated under some of these agreements to pay a percentage of any future sublicensing revenues that we may receive. Future license agreements may also include payment obligations such as milestone payments or minimum expenditures for research and development. We expect that any future licenses would contain reporting, insurance and indemnification requirements. We are actively reviewing and preparing additional patent applications to expand our patent portfolio, but there can be no assurances that patents related to our existing patent applications or any applications we may file in the future will be issued or that any issued patents will provide meaningful protection for our drug candidates, which could materially adversely affect our competitive business position, business prospects and financial condition.
Confidentiality agreements with employees and others may not adequately prevent disclosure of trade secrets and other proprietary information and may not adequately protect our intellectual property.
We rely on trade secrets to protect our technology, especially where we do not believe patent protection is appropriate or obtainable. However, trade secrets are difficult to protect. In order to protect our proprietary technology and processes, we also rely, in part, on confidentiality and intellectual property assignment agreements with our corporate partners, employees, consultants, outside scientific collaborators and sponsored researchers and other advisors. These agreements may not effectively prevent disclosure of confidential information nor result in the effective assignment to us of intellectual property, and may not provide an adequate remedy in the event of unauthorized disclosure of confidential information or other breaches of the agreements. In addition, others may independently discover our trade secrets and proprietary information, and in such case we could not assert any trade secret rights against such party. Enforcing a claim that a party illegally obtained and is using our trade secrets is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, courts outside the U.S. may be less willing to protect trade secrets. Costly and time-consuming litigation could be necessary to seek to enforce and determine the scope of our proprietary rights, and failure to obtain or maintain trade secret protection could materially adversely affect our business and financial condition.
| 32 |
Risks Related to Ownership of Our Securities
Because Vininder Singh, our Chief Executive Officer and director, controls a significant number of shares of our voting capital stock, he has significant influence over actions requiring stockholder approval.
Mr. Vininder Singh, our Chief Executive Officer and a director, beneficially owns approximately 18.7% of the Company’s common stock. As a result, Mr. Singh possesses significant influence on the outcome of matters submitted to our stockholders for approval, including the election of directors and any merger, consolidation or sale of all or substantially all of our assets. Accordingly, any investors who purchase shares will be minority stockholders and as such will have little to no say in the direction of us and the election of directors. Additionally, this concentration of ownership might harm the market price of our common stock by:
| ● | delaying, deferring or preventing a change in corporate control; | |
| ● | impeding a merger, consolidation, takeover or other business combination involving us; or | |
| ● | discouraging a potential acquirer from making a tender offer or otherwise attempting to obtain control of us. |
The price of our common stock may fluctuate substantially.
You should consider an investment in our common stock to be risky, and you should invest in our common stock only if you can withstand a significant loss and wide fluctuations in the market value of your investment. Some factors that may cause the market price of our common stock to fluctuate, in addition to the other risks mentioned in this “Risk Factors” section and elsewhere in this annual report, are:
| ● | sales of our common stock by our stockholders, executives, and directors; | |
| ● | volatility and limitations in trading volumes of our shares of common stock; | |
| ● | our ability to obtain financing to conduct and complete research and development activities; | |
| ● | our ability to attract new customers; | |
| ● | changes in the development status of the drugs we acquire; | |
| ● | failures to meet external expectations or management guidance; | |
| ● | changes in our capital structure or dividend policy or future issuances of securities; | |
| ● | our cash position; | |
| ● | announcements and events surrounding financing efforts, including debt and equity securities; | |
| ● | reputational issues; | |
| ● | announcements of acquisitions, partnerships, collaborations, joint ventures, new products, capital commitments, or other events by us or our competitors; | |
| ● | changes in general economic, political and market conditions in any of the regions in which we conduct our business; | |
| ● | changes in industry conditions or perceptions; | |
| ● | changes in valuations of similar companies or groups of companies; | |
| ● | analyst research reports, recommendations and changes in recommendations, price targets, and withdrawals of coverage; | |
| ● | departures and additions of key personnel; | |
| ● | disputes and litigations related to intellectual property rights, proprietary rights, and contractual obligations; | |
| ● | changes in applicable laws, rules, regulations, or accounting practices and other dynamics; and | |
| ● | other events or factors, many of which may be out of our control. |
In addition, if the market for stocks in our industry or industries related to our industry, or the stock market in general, experiences a loss of investor confidence, the trading price of our common stock could decline for reasons unrelated to our business, financial condition and results of operations. If any of the foregoing occurs, it could cause our stock price to fall and may expose us to lawsuits that, even if unsuccessful, could be costly to defend and a distraction to management.
We do not currently intend to pay dividends on our common stock and, consequently, investors’ ability to achieve a return on their investment will depend on appreciation in the price of our common stock.
We have never declared or paid cash dividends on our common stock. We currently intend to retain all of our future earnings, if any, to finance the growth and development of our business. As a result, capital appreciation, if any, of our common stock will be our stockholders’ sole source of gain for the foreseeable future. There is no guarantee that the price of our common stock will appreciate or remain equal to or above the price at which investors have purchased it.
| 33 |
If equity research analysts do not publish research or reports about our business or if they issue unfavorable commentary or downgrade our common stock, the price of our common stock could decline.
The trading market for our common stock may be affected by the research and reports that equity research analysts publish about us and our business. We do not control these analysts. The price of our common stock could decline if one or more equity analysts downgrade our common stock or if analysts issue other unfavorable commentary or cease publishing reports about us or our business.
Provisions of our charter documents or Nevada law could delay or prevent an acquisition of our company, even if the acquisition would be beneficial to our stockholders, and could make it more difficult to change management.
Provisions of our articles of incorporation and bylaws may discourage, delay or prevent a merger, acquisition or other change in control that stockholders might otherwise consider favorable, including transactions in which stockholders might otherwise receive a premium for their shares. In addition, these provisions may frustrate or prevent any attempt by our stockholders to replace or remove our current management by making it more difficult to replace or remove our board of directors. These provisions include:
| ● | limitations on our stockholders’ ability to call special meetings of stockholders; | |
| ● | an advance notice requirement for stockholder proposals and nominations for members of our Board; | |
| ● | the authority of our Board to determine the number of director seats on our Board; | |
| ● | the authority of our Board to fill vacancies occurring on the Board; and | |
| ● | the authority of our Board to issue preferred stock with such terms as our Board may determine. |
Our articles of incorporation grants our Board of Directors the power to designate and issue additional shares of common and/or preferred stock.
Our authorized capital consists of 100,000,000 shares of common stock and 10,000,000 shares of preferred stock. Our preferred stock may be designated into series pursuant to authority granted by our articles of incorporation, and on approval from our Board of Directors. The Board of Directors, without any action by our stockholders, may designate and issue shares in such classes or series as the Board of Directors deems appropriate and establish the rights, preferences and privileges of such shares, including dividends, liquidation and voting rights. The rights of holders of other classes or series of stock that may be issued could be superior to the rights of holders of our common stock. The designation and issuance of shares of capital stock having preferential rights could adversely affect other rights appurtenant to shares of our common stock.
We will indemnify and hold harmless our officers and directors to the maximum extent permitted by Nevada law.
Our bylaws provide that we will indemnify and hold harmless our officers and directors against claims arising from our activities, to the fullest extent not prohibited by Nevada law. If we were called upon to perform under our indemnification agreement, then the portion of our assets expended for such purpose would reduce the amount otherwise available for our business.
We are an “emerging growth company” under the JOBS Act of 2012 and we cannot be certain if the reduced disclosure requirements applicable to emerging growth companies will make our common stock less attractive to investors.
We are an “emerging growth company,” as defined in the Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”), and we may take advantage of certain exemptions from various reporting requirements that are not applicable to other public companies that are not “emerging growth companies” including, but not limited to, not being required to comply with the auditor attestation requirements of section 404 of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved. We cannot predict if investors will find our common stock less attractive because we may rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may be more volatile.
| 34 |
In addition, Section 107 of the JOBS Act also provides that an “emerging growth company” can take advantage of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act of 1933 (the “Securities Act”) for complying with new or revised accounting standards. In other words, an “emerging growth company” can delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We are choosing to take advantage of the extended transition period for complying with new or revised accounting standards.
We will remain an “emerging growth company” until the last day of the fiscal year following the fifth anniversary of the date of the first sale of our common stock pursuant to an effective registration statement under the Securities Act, although we will lose that status sooner if our revenues exceed $1.235 billion, if we issue more than $1 billion in non-convertible debt in a three year period, or if the market value of our common stock that is held by non-affiliates exceeds $700 million as of the last day of our most recently completed second fiscal quarter.
Investors may be unable to compare our business with other companies in our industry if they believe that our financial accounting is not as transparent as other companies in our industry. If we are unable to raise additional capital as and when we need it, our financial condition and results of operations may be materially and adversely affected.
We will continue to incur significant costs to ensure compliance with United States corporate governance and accounting requirements.
We will continue to incur significant costs associated with our public company reporting requirements, including costs associated with applicable corporate governance requirements such as those required by the Sarbanes-Oxley Act of 2002, and with other rules issued or implemented by the SEC. We expect all of these applicable rules and regulations to result in significant legal and financial compliance costs and to make some activities more time consuming and costly. We are currently evaluating and monitoring developments with respect to these rules, and we cannot predict or estimate the amount of additional costs we may incur or the timing of such costs.
If we fail to successfully maintain an effective internal control over financial reporting, the integrity of our financial reporting could be compromised, which could result in a material adverse effect on our reported financial results.
If we fail to maintain an effective system of internal control over financial reporting, we may not be able to accurately report our financial results or prevent fraud. As a result, stockholders could lose confidence in our financial and other public reporting, which would harm our business and the trading price of our common stock. Effective internal controls over financial reporting are necessary for us to provide reliable financial reports and, together with adequate disclosure controls and procedures, are designed to prevent fraud. Any failure to implement the required new or improved controls, or difficulties encountered in their implementation could cause us to fail to meet our reporting obligations. Inferior internal controls could also cause investors to lose confidence in our reported financial information, which could have a negative effect on the trading price of our common stock.
ITEM 1B. UNRESOLVED STAFF COMMENTS
Not applicable.
| 35 |
ITEM 1C. CYBERSECURITY
We conduct periodic risk assessments to identify cybersecurity threats, as well as assessments in the event of a material change in our business practices that may affect information systems that are vulnerable to such cybersecurity threats. These risk assessments include identification of reasonably foreseeable internal and external risks, the likelihood and potential damage that could result from such risks, and the sufficiency of existing policies, procedures, systems, and safeguards in place to manage such risks.
Following these risk assessments, we re-design, implement, and maintain reasonable safeguards to minimize identified risks; reasonably address any identified gaps in existing safeguards; and regularly monitor the effectiveness of our safeguards. Primary responsibility for assessing, monitoring and managing our cybersecurity risks rests with our Chief Information Officer, who reports to our Chief Executive Officer, to manage the risk assessment and mitigation process.
Our
cybersecurity incident response and vulnerability management policies are designed to escalate certain cybersecurity incidents to members
of management depending on the circumstances, including our Chief Executive Officer and Chief Financial Officer.
ITEM 2. PROPERTIES
Our principal business address is 325 Ellington Blvd, Unit 317, Gaithersburg, MD 20878, and our telephone number is 240-658-6710. Currently, we do not own any real property. All of our employees work virtually.
| 36 |
ITEM 3. LEGAL PROCEEDINGS
We are not currently a party to any legal or administrative proceedings. Our current officers and directors have not been convicted in criminal proceedings, nor have they been permanently or temporarily enjoined, barred, suspended, or otherwise limited from involvement in any type of business, securities or banking activities.
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable.
PART II
ITEM 5. MARKET FOR COMMON EQUITY AND RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
Information with Respect to our Common Stock and Tradeable Warrants
Our common stock is publicly traded on the Nasdaq Capital Market, or Nasdaq, and began trading under the symbol “BFRG” on February 14, 2023. Our tradeable warrants are traded on Nasdaq and began trading under the symbol “BFRGW” on February 14, 2023.
Holders of Record
As of the date of this filing, we had 21 shareholders of record of our common stock. This number does not include beneficial owners whose shares are held by nominees in street name.
Dividend Policy
Holders of common stock are entitled to receive ratably such dividends, if any, as may be declared by the Board of Directors out of funds legally available. We have not paid any dividends since our inception, and we presently anticipate that all earnings, if any, will be retained for the development of our business. Any future disposition of dividends will be at the discretion of our Board of Directors and will depend upon, among other things, our future earnings, operating and financial condition, capital requirements, and other factors.
Recent Sales of Unregistered Securities
Other than as previously reported in the Company’s Current Reports on Form 8-K, there were no unregistered sales of equity securities during the year ended December 31, 2025.
Securities Authorized for Issuance under Equity Compensation Plans
The information required by this item with respect to securities authorized for issuance under equity compensation plans is set forth in Part III, Item 12 of this Annual Report on Form 10-K, and is incorporated herein by reference.
Issuer Purchases of Equity Securities
We did not repurchase any of our equity securities during the year ended December 31, 2025.
ITEM 6. [RESERVED]
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS AND PLAN OF OPERATION
The following discussion and analysis of the results of operations and financial condition of BullFrog AI Holdings, Inc. (“BullFrog” or the “Company”) as of and for the years ended December 31, 2025 and 2024 should be read in conjunction with our consolidated financial statements and the notes to those consolidated financial statements that are included elsewhere in this Annual Report on Form 10-K. References in this Management’s Discussion and Analysis of Financial Condition and Plan of Operation to “us”, “we”, “our” and similar terms refer to the Company. This Management’s Discussion and Analysis of Financial Condition and Plan of Operation contains statements that are forward-looking. These statements are based on current expectations and assumptions that are subject to risk, uncertainties, and other factors. These statements are often identified by the use of words such as “may,” “will,” “expect,” “believe,” “anticipate,” “intend,” “could,” “estimate,” or “continue,” and similar expressions or variations. Actual results could differ materially because of the factors discussed in “Risk Factors” elsewhere in this Annual Report on Form 10-K, and other factors that we may not know.
| 37 |
OVERVIEW
BullFrog AI Holdings, Inc. was incorporated in the State of Nevada in February 2020. BullFrog AI Holdings, Inc. is the parent company of BullFrog AI, Inc. and BullFrog AI Management, LLC, which were incorporated in Delaware and Maryland, in 2017 and 2021, respectively. Operations are currently conducted through BullFrog AI Holdings, Inc., which began operations in February 2020. We are a company focused specifically on advanced Artificial Intelligence / Machine Learning (“AI/ML”) analysis of complex data in the advancement of medicine. Our AI/ML platform (trade name: bfLEAP™) was created from technology originally developed at The Johns Hopkins University Applied Physics Laboratory (“JHU-APL”). Subsequently, we have developed new tools and capabilities composed of an ensemble of machine learning and artificial intelligence models.
In February 2018, we secured an original exclusive, worldwide, royalty-bearing license from JHU-APL for the technology underlying our bfLEAP™ platform. The license covers three (3) issued patents, one (1) new provisional patent application, non-patent rights to proprietary libraries of algorithms and other trade secrets including modifications and improvements. We entered into a license agreement in July 2022 that provides the Company with new intellectual property and also encompasses most of the intellectual property from the February 2018 license. Our objective is to utilize bfLEAP™, our AI/ML platform, with a precision medicine approach toward drug development with biopharmaceutical collaborators, as well as with our own internal clinical development programs. We believe the bfLEAP™ platform is ideally suited for evaluating pre-clinical and clinical trial data generated in translational research and clinical trial settings in order to lead to faster, less expensive drug approvals.
Our aim is to improve the odds of success in each stage of developing medicine, ranging from early pre-clinical through late-stage clinical development. Our ultimate objective is to utilize bfLEAP™ to enable the success of ongoing third-party clinical trials or rescue late-stage failed drugs (i.e., Phase II or Phase III clinical trial failures) for in-house development and divestiture. We will also consider collaborations for earlier stage drugs.
In July 2022, we entered into an exclusive, worldwide, royalty-bearing license from JHU-APL that provides additional intellectual property rights including patents, copyrights, and knowhow for the technology underlying our bfLEAP™ analytical AI/ML platform. In consideration for the new license entered into in July 2022 with JHU-APL, we issued to JHU-APL 39,879 shares of common stock. Under the terms of the new license agreement, JHU-APL will be entitled to eight (8%) percent of net sales for the services provided by the Company to other parties and three (3%) percent for internally developed drug projects in which the JHU-APL license was utilized. The new license also contains tiered sub licensing fees that start at fifty (50%) percent and reduce to twenty-five (25%) percent based on revenues. In May 2023, we entered into Amendment Number 1 of the July 2022 license agreement with JHU-APL pursuant to which we gained access to certain improvements including additional patents and knowhow in exchange for a series of payments totaling $275,000. The first of these payments of $75,000 was paid in July 2023, the second of these payments was paid in June 2025, and the remaining payments of $75,000 and $50,000 are due in years 2026 and 2027, respectively. The amendment also reduced the 2023 minimum annual royalty payment from $80,000 to $60,000. All other financial terms remain the same. As a result of this amendment, the minimum annual payments were $30,000 for 2022 and $60,000 for 2023, and the minimum annual payments will be $300,000 for 2024 and beyond, all of which are creditable against royalties paid by us. As of December 31, 2025, we have accrued $300,000 for the 2025 minimum annual royalty payments and the entire accrued balance remains uninvoiced and unpaid as of the date of this filing.
We intend to continue to evolve and improve bfLEAP™, either in-house or with development partners like JHU-APL. We plan to leverage our proprietary AI/ML platform developed over several years at one of the top innovation institutions in the world, which has already been successfully applied in multiple sectors.
| 38 |
We operate and have staffed our business using funds from our initial public offering and subsequent financings. Since our incorporation, we have entered into various partnerships and relationships, completed our first commercial service contract with a leading rare disease non-profit organization for AI/ML analysis of late-stage clinical data in 2023, and completed our collaboration agreement for clinical trial optimization with a Phase III oncology company focused on novel chemotherapeutic treatments for rare cancers in the third quarter of 2025. We have also acquired the rights to a series of preclinical and early clinical drug assets from universities, as well as a strategic collaboration with a world-renowned research institution to create a HSV1 viral therapeutic platform to engineer immunotherapies for a variety of diseases. We have signed exclusive worldwide license agreements with JHU for a cancer drug that targets glioblastoma (brain cancer), pancreatic cancer, and others. We have also signed an exclusive worldwide license from GWU for another cancer drug that targets hepatocellular carcinoma (liver cancer) and other liver diseases. In addition, we have signed three-year strategic data and commercialization agreements with the Lieber Institute for Brain Development (“LIBD”) whom we believe has a repository of the largest collection of postmortem brains in the world, including molecular, clinical, and other data. The objective of this collaboration with LIBD is for the Company to analyze these rich data sets using its proprietary AI/ML tools and models and then go to market with the discoveries with the ultimate goal of securing revenue generating strategic partnership deals with biopharmaceutical companies. We intend to secure the rights to other proprietary data sets and repeat this strategy. Additionally, we intend to gain access to later-stage clinical assets through partnerships or the acquisition of rights to failed therapeutic candidates for drug rescue. In certain circumstances, we intend to conduct late-stage clinical trials in an effort to rescue therapeutic assets that previously failed. In these cases, there will be a requirement for drug supply and regulatory services to conduct clinical trials. The success of our clinical development programs will require finding partners to support the clinical development, adequate availability of raw materials and/or drug product for our R&D and clinical trials, and, in some cases, may also require establishment of third-party arrangements to obtain finished drug product that is manufactured appropriately under good manufacturing practices, and packaged for clinical use or sale. Since we are a company focused on using our AI/ML technology to advance medicines, any clinical development programs will also require, in all cases, partners and the establishment of third-party relationships for execution and completion of clinical trials.
Since completing our initial public offering in February 2023 (the “IPO”), aided by the receipt of the IPO proceeds in addition to the proceeds from our February 2024 and October 2024 offerings and our ongoing At-The-Market Sales Agreement with BTIG, LLC (the “ATM Agreement”) and common stock purchase agreement with Lincoln Park Capital Fund, LLC, we have implemented several initiatives including: investor relations and marketing to raise awareness of the Company in the financial and business sectors, research and development, and initiation of preclinical studies with our in-licensed drug programs. The Company is actively engaged in developing and pursuing new intellectual property as it strives to continuously evolve its AI/ML platform.
Internally, we have added incremental staff to accelerate execution and the development of processes and custom scripts for use in performing new drug target discovery and analytical services for customers, while also launching initiatives targeting large public health data sources and seeking access to proprietary health data sources, such as our agreement with the LIBD. We are also continuing to improve our accounting and financial reporting systems and processes to enhance our internal control environment as a public company. Capital from the IPO was also used to retire two notes that were sold to fund the Company through the IPO as well as other debts accrued over time to our staff, employees and consultants, and obligations related to the acquisition of our licensed drug programs.
We have had negative cash flows from operations and operated at a net loss since inception. In the first quarter of 2023, we completed our IPO. In February 2024, we received net proceeds of approximately $5.7 million from an underwritten public offering of common stock and warrants. In October 2024, we received net proceeds of approximately $2.7 million from a registered direct offering of common stock and pre-funded warrants, and concurrent private placement of common stock warrants. Through December 31, 2025, we received approximately $2.6 million of net proceeds from the sale of our common stock pursuant to the ATM Agreement. As of December 31, 2025, the Company has a cash balance of approximately $2.3 million, which includes restricted cash of $0.1 million held by a financial institution as collateral for the Company’s corporate credit card program. As of December 31, 2025, the Company’s cash and cash equivalents position is not sufficient to fund the Company’s planned operations for at least a year beyond the filing date of the consolidated financial statements. These factors raise substantial doubt about the Company’s ability to continue as a going concern. The ability to continue as a going concern is dependent upon us utilizing the financing facilities available to us and/or obtaining necessary additional financing and/or revenues to meet our obligations arising from normal business operations when they become due.
| 39 |
Our Strategy
We have a strategy designed to reduce risk and increase the frequency of cash flow. The first part of the strategy is to generate revenues through strategic relationships with biopharma companies. These relationships will be structured as a combination of fees in cash and, in some instances, equity in our partners, or other consideration and intellectual property based on the specific scope of the engagement. The objective of these engagements will be to uncover valuable insights to reduce the risk and increase the speed of the drug development process, which can be achieved through manual or automated integration into the client’s workflow or analysis of discrete data sets.
In the future, the second part of our strategy involves acquiring the rights to drugs at various stages of development and using our proprietary AI/ML technology to advance the development of such drugs, with the objective of creating near term value and then exiting and monetizing as quickly as possible, preferably within approximately 30 months.
Results of Operations
For the years ended December 31, 2025 and 2024
Collaboration Revenue and Cost of Collaboration Revenue
In the year ended December 31, 2025, we recognized revenue and cost of revenue of approximately $117,000 and $95,000, respectively, which entirely related to our lone collaboration agreement with Eleison Pharmaceuticals Inc. (“Eleison”), a Phase III oncology company focused on novel chemotherapeutic treatments for rare cancers. We had no active customer agreements in the year ended December 31, 2024 and, therefore, no revenue was recognized.
Operating Expenses
| Year ended December 31, | Net Change | |||||||||||||||
| 2025 | 2024 | $ | % | |||||||||||||
| Operating expenses: | ||||||||||||||||
| Research and development | $ | 1,799,738 | $ | 2,223,265 | $ | (423,527 | ) | -19 | % | |||||||
| General and administrative | 4,816,790 | 5,013,118 | (196,328 | ) | -4 | % | ||||||||||
| Total operating expenses | $ | 6,616,528 | $ | 7,236,383 | $ | (619,855 | ) | -9 | % | |||||||
Research and Development
Our research and development expenses for the year ended December 31, 2025 decreased compared the year ended December 31, 2024, primarily due to a reduction in personnel costs and the allocation of certain personnel costs from research and development to cost of revenue related to our collaboration with Eleison.
General and Administrative
Our general and administrative expenses for the year ended December 31, 2025 decreased compared to the year ended December 31, 2024, primarily due to reductions in our director and officer insurance policy premium and recruiting fees, partially offset by an increase in noncash stock-based compensation expense.
Other Income (Expense), Net
Interest expense decreased by approximately $12,000 for the year ended December 31, 2025, compared to the same period ended December 31, 2024 due to a decrease in our director and officer insurance policy premium loan. Interest income decreased by approximately $159,000 primarily due to a decrease in our average cash balances.
Liquidity and Capital Resources
Through December 31, 2025, we have an accumulated deficit of approximately $23.3 million and we have funded our operations through the sale of common stock, warrants and debt. We anticipate that our expenses will increase in the future to support our service offerings, clinical and pre-clinical research and development activities associated with strategic partnering and collaborations, as well as acquired product candidates. These increases could include increased costs related to the hiring of additional personnel and fees to outside consultants, lawyers, and accountants, among other expenses.
| 40 |
In February 2024, we completed an underwritten offering of common stock and warrants generating approximately $5.7 million of net proceeds. In October 2024, we completed a registered direct offering of common stock and pre-funded warrants, and concurrent private placement of common stock warrants generating approximately $2.7 million of net proceeds.
In April 2025, we entered into an ATM Agreement with BTIG, LLC, pursuant to which we may offer and sell shares of common stock, from time to time in our sole discretion, at the market price up to an aggregate offering price of $20 million. We are not obligated to sell any shares, and BTIG is not required to sell any specific number or dollar amount of shares of common stock. Accordingly, we will not receive any proceeds from such transaction until shares are actually sold by BTIG. Subject to our request to sell shares, BTIG will use commercially reasonable efforts, consistent with its normal trading and sales practices, to sell shares of common stock on our behalf in accordance with Company instructions. Notwithstanding the foregoing, there can be no assurance that we will be able to sell, when needed, sufficient shares under the ATM Agreement to fund planned operations. In the year ended December 31, 2025, we received approximately $2.6 million of net proceeds from the sale of 1,686,511 shares of our common stock at an average price of approximately $1.59 per share. Subsequent to year end 2025, we received approximately $0.9 million of net proceeds from the sale of 976,204 shares of our common stock at an average price of approximately $0.90 per share. Consequently, as of the date of this filing, approximately $16.4 million of capacity remains available under the ATM Agreement; however, the amount we are permitted to raise in any 12-month period is currently limited based on our public float pursuant to SEC General Instruction I.B.6 of Form S-3. Accordingly, as of the date of this filing, we are limited to additional common stock sales of approximately $1.1 million under the ATM Agreement.
In September 2025, we entered into a purchase agreement with Lincoln Park Capital Fund, LLC (“Lincoln Park”), pursuant to which Lincoln Park committed to purchase up to $10.0 million of our common stock, subject to certain limitations. We have the right, but not the obligation, to sell to Lincoln Park, and Lincoln Park is obligated to purchase up to $10.0 million of our common stock. Such sales of common stock by the Company, if any, will be subject to certain limitations set forth in the purchase agreement, and may occur from time to time, at our sole discretion, over the 36-month period commencing on November 25, 2025, the date that the conditions to Lincoln Park’s purchase obligation set forth in the purchase agreement were satisfied. In connection with the purchase agreement, we issued 147,682 shares of common stock valued at approximately $207,000 to Lincoln Park as a fee in advance of any sales pursuant to this facility. No shares were sold under this facility during the year ended December 31, 2025. In January 2026, we received net proceeds of approximately $218,000 from the sale of 270,000 shares of common stock at an average price of approximately $0.81 per share.
As of December 31, 2025, our cash and cash equivalents position is not sufficient to fund our planned operations for at least a year beyond the filing date of the consolidated financial statements. These factors raise substantial doubt about our ability to continue as a going concern. The ability to continue as a going concern is dependent upon us utilizing the financing facilities available to us and/or obtaining necessary additional financing and/or revenues to meet our obligations arising from normal business operations when they become due. Accordingly, we will seek additional capital to continue to execute our strategy as discussed above.
On August 21, 2025, we received a letter from the listing staff of The Nasdaq Stock Market LLC (“Nasdaq”) that we were no longer in compliance with the minimum stockholders’ equity requirement for continued listing on Nasdaq pursuant to Nasdaq Listing Rule 5550(b)(1) (the “Stockholders’ Equity Rule”). The Stockholders’ Equity Rule requires companies listed on the Nasdaq Capital Market to maintain stockholders’ equity of at least $2,500,000 or to meet alternatives of market value of listed securities or net income from continuing operations, which we do not currently meet. In accordance with Nasdaq rules, we had 45 calendar days, or until October 6, 2025, to submit a plan to regain compliance. After submitting the plan to regain compliance, on October 7, 2025, Nasdaq granted us an extension until February 17, 2026, to comply with Listing Rule 5550(b)(1). On February 19, 2026, we received a further notice from Nasdaq (the “February Letter”) notifying us that Nasdaq determined that we had not met the terms of the extension. We thereafter timely requested a hearing before an independent Nasdaq Hearings Panel (the “Panel”) which automatically stayed any suspension or delisting action pending the hearing and the expiration of any extension period granted by the Panel following the hearing. At the hearing, we plan to present additional details of our Plan and provide an update on our efforts to regain compliance. We will also request additional time to complete the steps of our Plan and regain compliance with all applicable Nasdaq Listing Rules.
| 41 |
On February 10, 2026, we received a letter from Nasdaq notifying us that, for the last 30 consecutive business days, the closing bid price for our common stock, par value $0.00001 per share (the “Common Stock”), was below $1.00 per share, which is the minimum closing bid price required for continued listing on the Nasdaq Global Market (the “Minimum Bid Price Requirement”) pursuant to Nasdaq Listing Rule 5550(a)(2) (the “Bid Price Notice”). The Bid Price Notice had no immediate effect on the listing of our Common Stock and tradeable warrants. As such, our Common Stock will continue to trade on the Nasdaq Capital Market under the symbol “BFRG,” and our tradeable warrants will continue to trade on the Nasdaq Capital Market under the symbol “BFRGW.” In accordance with Nasdaq Listing Rule 5810(c)(3)(A), we were provided a compliance period of 180 calendar days from the date of the Bid Price Notice, or until August 10, 2026, to regain compliance with the Minimum Bid Price Requirement. If at any time during the 180-calendar day grace period, the closing bid price of our Common Stock is at least $1.00 per share for a minimum of ten consecutive business days (unless the Nasdaq staff exercises its discretion to extend this ten business day period pursuant to Nasdaq Listing Rule 5810(c)(3)(H)), Nasdaq will provide us written confirmation of compliance, and the matter will be closed. If we do not regain compliance during the initial 180-calendar day compliance period, we may be provided a second 180-calendar day period to regain compliance. If we do not regain compliance within the allotted compliance periods, including any extensions that may be granted by Nasdaq, our listed securities will be subject to delisting. We would thereafter have the right to appeal a determination to delist our securities, and our securities would remain listed on the Nasdaq Capital Market until the completion of the appeal process. Notably, at a Special Meeting of Stockholders in October 2025, the Company received stockholder approval to effect a reverse stock split at a ratio of not less than 1-to-2 and not more than 1-to-15, such ratio and timing to be determined in the discretion of the Company’s Board of Directors.
Consolidated Cash Flow Data
| Year ended December 31, | ||||||||||||
| 2025 | 2024 | Net Change | ||||||||||
| Net cash (used in) provided by | ||||||||||||
| Operating activities | $ | (5,522,265 | ) | $ | (5,610,249 | ) | $ | 87,984 | ||||
| Investing activities | - | - | - | |||||||||
| Financing activities | 2,374,987 | 8,421,502 | (6,046,515 | ) | ||||||||
| Net (decrease) increase in cash and cash equivalents | $ | (3,147,278 | ) | $ | 2,811,253 | $ | (5,958,531 | ) | ||||
Cash Flows Used in Operating Activities
Net cash used in operating activities for the year ended December 31, 2025 decreased by approximately $88,000 compared to the year ended December 31, 2024, primarily due to decreased operating costs in 2025 attributable to a reduction in personnel costs, partially offset by the timing and payment of vendor invoices and associated impact to accounts payable.
Cash Flows Used in Investing Activities
There was no cash used in investing activities during any of the periods presented.
Cash Flows Provided by Financing Activities
Net cash provided by financing activities for the year ended December 31, 2025 decreased, compared to the year ended December 31, 2024, primarily due to proceeds from our offerings and warrant exercises in 2024, partially offset by proceeds from sales of common stock under our ATM Agreement in 2025.
Critical Accounting Policies
In Note 2 of our Audited Financial Statements for the year ended December 31, 2025 found elsewhere in this Annual Report on Form 10-K, we included a discussion of the most critical accounting policies used in the preparation of our financial statements.
Off-Balance Sheet Arrangements
We do not have any off-balance sheet arrangements, as such term is defined in Item 303(a)(4) of Regulation S-K.
| 42 |
Financial Operations Overview
Revenue
In February 2025, we entered into a collaboration agreement with Eleison Pharmaceuticals Inc., a Phase III oncology company focused on novel chemotherapeutic treatments for rare cancers, and we recognized revenue of approximately $117,000 pursuant to this agreement. Additionally, in June 2025, we entered into a strategic collaboration agreement with Sygnature Discovery (“Sygnature”), pursuant to which we established a joint marketing arrangement where Sygnature will introduce our BullFrog Data Networks™ platform to Sygnature’s global biopharma client base; however, we have not yet recognized any revenue under this collaboration. While we are currently in discussions with other potential partners, there can be no assurance of entering into other business relationships. We did not recognize any revenue in 2024.
Cost of Revenue
Cost of revenue consists primarily of the allocation of personnel costs (e.g. payroll, benefits, and consulting fees) of our employees and third-party consultants directly attributable to the satisfaction of our performance obligations under our revenue arrangements.
Research and Development Costs and Expenses
Research and development costs and expenses include development activities related to our licensed drug candidates and our discovery efforts and collaborations. In addition to fees paid to external service providers, we are also allocating costs for internal personnel working on these activities as well as their efforts to develop our product and service offerings using bfLEAP™. We anticipate that our research and development costs could become significant over time as we execute on our business plan and begin conducting preclinical research and development activities directed at securing development partners and filing an investigational new drug (IND) application for our licensed drug development programs described in this filing, as well as under strategic partnerships and for other drug development programs we may acquire. Research and development expenses are recorded in operating expenses in the period in which they are incurred. Estimates will be used in determining the expense liability of certain costs where services have been performed but not yet invoiced. We will monitor levels of performance under each significant contract for external services through communications with the service providers to reflect the actual amount expended.
General and Administrative Expenses
General and administrative costs and expenses include personnel costs and costs associated with being a public company such as directors and officers insurance, audit and tax provider fees, legal fees, and exchange listing costs. Additionally, our general and administrative costs include expenses for our business development, investor relations and marketing efforts. We anticipate our general and administrative expenses increasing in the future to support our service offerings and clinical and pre-clinical research and development activities associated with strategic partnering and collaborations.
Emerging Growth Company and Smaller Reporting Company Status
We are an emerging growth company as defined in the Jumpstart Our Business Startups Act of 2012 (“JOBS Act”) and may take advantage of reduced reporting requirements that are otherwise applicable to public companies. Section 107 of the JOBS Act exempts emerging growth companies from being required to comply with new or revised financial accounting standards until private companies are required to comply with those standards. We have elected to use the extended transition period to comply with new or revised accounting standards. This may make it difficult to compare our financial results with the financial results of another public company that is either not an emerging growth company or is an emerging growth company that has chosen not to take advantage of the extended transition period exemptions because of the potential differences in accounting standards used.
We are also considered a “smaller reporting company”, meaning that the market value of our stock held by non-affiliates plus the aggregate amount of gross proceeds to us as a result of the IPO is less than $700 million and our annual revenue was less than $100 million during the most recently completed fiscal year. We may continue to be a smaller reporting company if either (i) the market value of our stock held by non-affiliates is less than $250 million or (ii) our annual revenue was less than $100 million during the most recently completed fiscal year and the market value of our stock held by non-affiliates is less than $700 million. If we are a smaller reporting company at the time we cease to be an emerging growth company, we may continue to rely on exemptions from certain disclosure requirements that are available to smaller reporting companies. Specifically, as a smaller reporting company we may choose to present only the two most recent fiscal years of audited financial statements in our Annual Report on Form 10-K and, similar to emerging growth companies, smaller reporting companies have reduced disclosure obligations regarding executive compensation.
| 43 |
ITEM 7A. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
As a smaller reporting company, we are not required to provide the information required by this Item.
ITEM 8. FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
The financial statements required to be filed pursuant to this Item 8 are appended to this Annual Report on Form 10-K. See “Index to Consolidated Financial Statements” which appears on page F-1 of this Annual Report on Form 10-K, and is incorporated herein by reference.
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
None.
ITEM 9A. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
We are required to maintain “disclosure controls and procedures” as such term is defined in Rule 13a-15(e) under the Securities Exchange Act of 1934 (the “Exchange Act”). In designing and evaluating our disclosure controls and procedures, our management recognized that disclosure controls and procedures, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of disclosure controls and procedures are met. The design of any disclosure controls and procedures is also based, in part, upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions. We conducted an evaluation of the effectiveness of our disclosure controls and procedures as of December 31, 2025. Based on this evaluation, our Chief Executive Officer and Chief Financial Officer concluded that our disclosure controls and procedures were effective as of the end of the reporting period covered in this Annual Report on Form 10-K.
Management’s Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as defined in Exchange Act Rule 13a-15. Internal control over financial reporting is defined in Rule 13a-15(f) and 15(d)-15(f) under the Exchange Act as a process designed to provide reasonable assurance to our management and Board of Directors regarding the preparation and fair presentation of published financial statements.
A control system, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the control system are met. Further, the design of a control system must reflect the fact that there are resource constraints, and the benefits of controls must be considered relative to their costs. Because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that all control issues and instances of error or fraud, if any, within our Company have been detected. However, these inherent limitations are known features of the financial reporting process. Therefore, it is possible to design safeguards into the process to reduce, though not eliminate, this risk.
Management conducted an assessment of our internal control over financial reporting as of December 31, 2025 based on the framework and criteria established by the Committee of Sponsoring Organizations of the Treadway Commission in Internal Control-Integrated Framework (2013). Based on the assessment, and the noted remediation of the previously identified material weaknesses, management concluded that, as of December 31, 2025, our internal control over financial reporting was effective.
| 44 |
Remediation of Previously Reported Material Weaknesses
As previously disclosed, management identified material weaknesses in its internal controls over financial reporting at December 31, 2023 which continued to be unremediated as of December 31, 2024. Specifically, Management noted the Company did not properly document, implement or operate a system of effective internal controls over financial reporting. A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of the company’s annual or interim financial statements will not be prevented or detected on a timely basis.
As of December 31, 2025, management implemented the following improvements to its internal controls over financial reporting. Namely, the Company:
| ● | transitioned its day-to-day accounting processes to an external firm including automating its vendor payments; | |
| ● | completed the transfer of the overall accounting process to an enterprise type accounting platform; | |
| ● | reviewed the design and effectiveness of our controls including the creation of an annual risk assessment and ongoing monitoring activities; and | |
| ● | evaluated all internal and external resources to ensure they are appropriate for the level and complexity of our current operations, including the hiring of a Corporate Controller in 2024. |
Management has evaluated these improvements and remediation efforts and believes its internal controls over financial reporting are now operating effectively, and consequently, determined that the material weaknesses have been remediated.
Changes in Internal Control Over Financial Reporting
Other than the changes noted above related to the remediation of material weaknesses, there has been no change in our internal controls over financial reporting during the most recently completed fiscal quarter that has materially affected, or is reasonably likely to materially affect, our internal controls over financial reporting.
ITEM 9B. OTHER INFORMATION
Insider Trading Plans
During
the quarter ended December 31, 2025, none of our directors or executive officers
ITEM 9C. DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS
Not applicable.
| 45 |
PART III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS, AND CORPORATE GOVERNANCE
Executive Officers and Directors
The following table sets forth the name, age and position of each of our executive officers, key employees and directors.
| Name | Age | Position(s) | ||
| Executive Officers: | ||||
| Vininder Singh | 58 | Chief Executive Officer and Director | ||
| Josh Blacher | 54 | Chief Financial Officer | ||
| Non-Executive Directors: | ||||
| R. Donald Elsey | 73 | Director and Chair of Audit Committee | ||
| William Enright | 63 | Director and Chair of Compensation Committee | ||
| Jason D. Hanson | 58 | Director and Chair of Nominating and Corporate Governance Committee |
Vininder (Vin) Singh is the Founder, Chairman, and Chief Executive Officer of BullFrog AI Holdings, Inc. since its inception in August 2017. Over the past eight years, he has built the Company from scratch and during that time he has led strategy, built a highly experienced team of leaders, spear headed the acquisition and development of BullFrog’s core AI technology and drug assets, secured the first revenue, and raised approximately $2 million in financing prior to the Company’s IPO in February 2023. In February 2020, he formed BullFrog AI Holdings, Inc., and BullFrog AI Inc. became a wholly owned subsidiary designated as the holder of core intellectual property. Mr. Singh is a serial entrepreneur and experienced executive with 25 years of experience in the life sciences and biotechnology industries. He has extensive start-up experience having founded and built several pioneering investor-backed companies including BullFrog AI, which uses machine learning/AI to enable drug development, Next Healthcare Inc., a personalized diagnostics and adult cell banking service, and MaxCyte Inc. (NASDAQ: MXCT), a cell therapy company. He was also an executive at GlobalStem Inc. and ThermoFisher Scientific, leading their global cell therapy services business. Mr. Singh has a B.S. in Electrical Engineering from Rutgers University, an M.S. in Biomedical Engineering from Rensselaer Polytechnic Institute, and an M.B.A. from Johns Hopkins University. We believe that Mr. Singh is qualified to serve as a member of our board of directors due to the perspective and experience that he brings as our Founder and Chief Executive Officer and his extensive experience in the science and biotechnology industries and in the management of startup companies.
Josh Blacher has been the Chief Financial Officer of BullFrog AI Holdings, Inc. since December 2024. Mr. Blacher has served as an employee of Danforth Advisors since September 2022, where he has worked as a Chief Financial Officer in a consulting capacity for a number of life sciences companies as well as Managing Partner of Columbus Circle Capital LLC since August 2019. In his capacity as a consultant of Danforth, he has served as Chief Financial Officer of Predictive Oncology since September 2023 as well as Axe Compute, among others. During his tenure at Columbus Circle Capital, Mr. Blacher has served as chief financial officer at several public and private companies. Prior to his tenure at Columbus Circle Capital, Mr. Blacher served as Chief Business Officer at InMed Pharmaceuticals (Nasdaq: INM) from April 2018 to August 2019, as Chief Financial Officer of Therapix Biosciences (Nasdaq: TRPX) from April 2017 to April 2018, and as Chief Financial Officer at Galmed Pharmaceuticals (Nasdaq: GLMD) from October 2014 to March 2017. Earlier in his career, Mr. Blacher served in senior capacities at Teva Pharmaceuticals, Deutsche Asset Management and Morgan Stanley. Mr. Blacher holds a Bachelor of Arts from Yeshiva University and an M.B.A. from Columbia Business School.
R. Don Elsey has been a director and chair of the Audit Committee of our board since February 14, 2023. Mr. Elsey was the CFO of Lyra Therapeutics from 2019 until his retirement in December 2020. Previously, from February 2015 to February 2019, Mr. Elsey served as Chief Financial Officer at Senseonics, Inc., a medical device company. From May 2014 until February 2015, Mr. Elsey served as Chief Financial Officer of Regado Biosciences, Inc., a biopharmaceutical company. From December 2012 to February 2014, Mr. Elsey served as Chief Financial Officer of LifeCell Corporation, a privately held regenerative medicine company. Mr. Elsey previously served as the chair of the Audit Committee and member of the Board of Directors of OpGen, Inc., a precision medicine company. Mr. Elsey holds a B.A. in economics and an M.B.A. in finance from Michigan State University. We believe that Mr. Elsey is qualified to serve as a member of our board of directors because of his extensive professional experience in science and biotechnology companies.
William “Bill” Enright has been a director and chair of the Compensation Committee of our board since February 14, 2023. He is a seasoned biotech executive with more than thirty-five years of experience in building and financing both privately held and publicly held companies and he is currently the Chief Executive Officer and a Director of Barinthus Biotherapeutics plc (NASDAQ: BRNS), which he helped take public in April 2021. Prior to Barinthus, Mr. Enright spent more than ten years at Altimmune (NASDAQ: ALT) as a Director, President & Chief Executive Officer, moving multiple programs into clinical testing, completing several acquisitions, and eventually taking the company public. Prior to joining Altimmune, Mr. Enright spent six years with GenVec, Inc. (acquired by Precigen) with increasing responsibilities, culminating as Head of Business Development. He brings a breadth of experience in a variety of positions within the life science/biotech industry, including time as a consultant, a bench scientist and 12 years with Life Technologies, Inc. (acquired by Thermo-Fisher), working in various senior level licensing, business management, manufacturing and research roles. Mr. Enright received a Master of Arts in Molecular Biology from SUNY at Buffalo and a Master of Science in Business Management from Johns Hopkins University. We believe that Mr. Enright is qualified to serve as a member of our board of directors because of his extensive professional experience in life science/biotech companies and in the management of public companies.
| 46 |
Jason Hanson has served as a director and chair of the Nominating and Corporate Governance Committee since February 14, 2023. Mr. Hanson served as Chief Executive Officer and as a Director of enGene Inc. from July 2018 to July 2024. He also served as President of enGene Inc. from July 2018 to December 2022. Mr. Hanson effectively launched enGene from a small private company working in the GI discovery space into a clinical stage gene therapy oncology company trading on Nasdaq, with a massively differentiated lead product to treat bladder cancer, implementing a new scientific, technical and strategic vision for the Company. From August 2016 to November 2017, Mr. Hanson served as President and Chief Executive Officer of Ohana Biosciences, a biotechnology company based in Cambridge, MA, and as a member of the Ohana Board of Directors and consultant to Ohana from November 2017 to June 2018. Mr. Hanson previously served as Executive Vice President and Chief Strategy Officer for NuVasive, Inc. from November 2015 to August 2016. Mr. Hanson served as Corporate Vice President of General Electric Company and member of the senior executive team of GE Healthcare, a global pharmaceutical, medical device and healthcare services business from May 2014 to October 2015. In January 2013, Mr. Hanson served as Company Group Chairman and Executive Vice President of Valeant Pharmaceuticals International, Inc. (now Bausch Health Companies Inc.). Previously, he served in various roles at Medicis Pharmaceutical Corporation, including as Executive Vice President and Chief Operating Officer between July 2006 and December 2012. Mr. Hanson also served in numerous roles at GE Healthcare, including General Counsel roles, from April 1999 to July 2006. Mr. Hanson holds a B.S. from Cornell University and a J.D. from Duke University School of Law. We believe that Mr. Hanson is qualified to serve as a member of our board of directors because of his extensive professional experience in science and biotechnology companies.
Role of Board of Directors in Risk Oversight Process
The board of directors has extensive involvement in the oversight of risk management related to us and our business and accomplishes this oversight through the regular reporting by the Audit Committee. The information set forth in Item 1C is incorporated herein by reference.
Director Independence
Messrs. Elsey, Enright and Hanson, three members of our Board of Directors, are independent using the definition of independence under Nasdaq Listing Rule 5605(a)(2) and the standards established by the SEC.
Committees of our Board
Audit Committee
Our audit committee consists of Don Elsey, William Enright and Jason Hanson, with Mr. Elsey serving as chair. Our board of directors has affirmatively determined that each meets the definition of “independent director” under the rules of The Nasdaq Capital Market, and that they each meet the independence standards under Rule 10A-3. Each member of our audit committee meets the financial literacy requirements of Nasdaq rules, and qualifies as a financial expert within the meaning of SEC regulations and meets the financial sophistication requirements of the pertinent listing standards of Nasdaq, as in effect from time to time. In making this determination, our board of directors has considered the members’ formal education and previous and current experience in financial roles. Our board of directors has adopted a written charter for the audit committee, which can be found on our website at https://ir.bullfrogai.com/corporate-governance/governance-documents.
The audit committee is appointed by the board of directors to assist the board of directors in its duty to oversee the Company’s accounting, financial reporting, and internal control functions and the audit of the Company’s financial statements. The role of the audit committee is to oversee management in the performance of its responsibility for the integrity of the Company’s accounting and financial reporting and its systems of internal controls, the performance and qualifications of the Company’s independent auditor, including the independent auditor’s independence, the performance of the Company’s internal audit function, and the Company’s compliance with legal and regulatory requirements. The audit committee met five times in 2025.
| 47 |
Compensation Committee
Our compensation committee consists of William Enright, Don Elsey and Jason Hanson, with Mr. Enright serving as chair. Our board of directors has adopted a written charter for the compensation committee, which can be found on our website at https://ir.bullfrogai.com/corporate-governance/governance-documents.
The compensation committee is responsible for reviewing and recommending, among other things:
| ● | the adequacy and form of compensation of the board; | |
| ● | the compensation of the Chief Executive Officer, including base salary, incentive bonus, stock option and other grant awards, and benefits upon hiring and on an annual basis; | |
| ● | the compensation of other senior management upon hiring and on an annual basis; and | |
| ● | the Company’s incentive compensation and other equity-based plans and recommending changes to such plans to our board of directors, when necessary. |
The compensation committee met two times in 2025.
Nominating & Corporate Governance Committee
Our nominating and corporate governance committee consists of Jason Hanson, William Enright and Don Elsey, with Mr. Hanson serving as chair. Our board of directors has adopted a written charter for the nominating and corporate governance committee, which can be found on our website at https://ir.bullfrogai.com/corporategovernance/governance-documents.
The nominating and corporate governance committee is responsible for, among other things:
| ● | developing criteria for membership on the board of directors and committees; | |
| ● | identifying individuals qualified to become members of the board of directors; | |
| ● | recommending persons to be nominated for election as directors and to each committee of the board of directors; | |
| ● | annually reviewing our corporate governance guidelines; and | |
| ● | monitoring and evaluating the performance of the board of directors and leading the board in an annual self-assessment of its practices and effectiveness. |
The nominating and corporate governance committee met one time in 2025.
Term of office
All directors hold office until the next annual meeting of the stockholders of the company and until their successors have been duly elected and qualified. Officers are elected by and serve at the discretion of our Board.
Code of Business Conduct and Ethics
We have adopted a Code of Business Conduct and Ethics that applies to our principal executive officer, principal financial officer, principal accounting officer or controller, employees and persons performing similar functions. Our code of ethics can be found at https://ir.bullfrogai.com/corporate-governance/governance-documents.
Clawback Policy
The Board adopted the BullFrog AI Compensation Recoupment Policy (the “Clawback Policy”), effective in December 2023, providing for the recovery of certain incentive-based compensation from current and former executive officers of the Company in the event the Company is required to restate any of its financial statements filed with the SEC under the Exchange Act in order to correct an error that is material to the previously-issued financial statements, or that would result in a material misstatement if the error were corrected in the current period or left uncorrected in the current period. Adoption of the Clawback Policy was mandated by new Nasdaq listing standards introduced pursuant to Exchange Act Rule 10D-1. The Clawback Policy is in addition to Section 304 of the Sarbanes-Oxley Act of 2002 which permits the SEC to order the disgorgement of bonuses and incentive-based compensation earned by a registrant issuer’s chief executive officer and chief financial officer in the year following the filing of any financial statement that the issuer is required to restate because of misconduct, and the reimbursement of those funds to the issuer. A copy of the Clawback Policy is attached as an exhibit to this annual report and can be found at https://ir.bullfrogai.com/corporate-governance/governance-documents.
| 48 |
Family Relationships
There are no family relationships among and between the issuer’s directors, officers, persons nominated or chosen by the issuer to become directors or officers, or beneficial owners of more than ten percent of any class of the issuer’s equity securities.
Involvement in Certain Legal Proceedings
Our directors and executive officers have not been involved in any of the following events during the past ten years:
| 1. | any bankruptcy petition filed by or against such person or any business of which such person was a general partner or executive officer either at the time of the bankruptcy or within two years prior to that time; |
| 2. | any conviction in a criminal proceeding or being subject to a pending criminal proceeding (excluding traffic violations and other minor offenses); |
| 3. | being subject to any order, judgment, or decree, not subsequently reversed, suspended or vacated, of any court of competent jurisdiction, permanently or temporarily enjoining him from or otherwise limiting his involvement in any type of business, securities or banking activities or to be associated with any person practicing in banking or securities activities; |
| 4. | being found by a court of competent jurisdiction in a civil action, the SEC or the Commodity Futures Trading Commission to have violated a Federal or state securities or commodities law, and the judgment has not been reversed, suspended, or vacated; |
| 5. | being subject of, or a party to, any Federal or state judicial or administrative order, judgment decree, or finding, not subsequently reversed, suspended or vacated, relating to an alleged violation of any Federal or state securities or commodities law or regulation, any law or regulation respecting financial institutions or insurance companies, or any law or regulation prohibiting mail or wire fraud or fraud in connection with any business entity; or |
| 6. | being subject of or party to any sanction or order, not subsequently reversed, suspended, or vacated, of any self-regulatory organization, any registered entity or any equivalent exchange, association, entity or organization that has disciplinary authority over its members or persons associated with a member. |
Section 16(a) Beneficial Ownership Compliance
Based solely upon a review of copies of such forms filed on Forms 3, 4 and 5, and amendments thereto furnished to us, we believe that as of the date of this Report, our executive officers, directors and greater than 10 percent beneficial owners have complied on a timely basis with all Section 16(a) filing requirements, except for one Form 4 by each of Messrs. Elsey, Enright and Hanson concerning the annual grant of stock options in September 2024.
Nomination Process
As of December 31, 2025, we did not affect any material changes to the procedures by which stockholders may recommend nominees to the Board of Directors.
Insider Trading Policies
We
have
| 49 |
ITEM 11. EXECUTIVE COMPENSATION
Summary Compensation Table
The following table sets forth all plan and non-plan compensation for the last two fiscal years paid to individuals who served as the Company’s principal executive officers and the Company’s two other most highly compensated executive officers serving as executive officers at the end of the last completed fiscal year, as required by Item 402(m)(2) of Regulation S-K of the Securities Act. We refer to these individuals collectively as our “named executive officers.”
| Name and Principal Position | Year | Salary | Bonus | Stock Awards[1] | Option Awards | All Other Compensation | Nonequity Incentive Plan Compensation | Nonqualified Deferred Compensation Earnings | Total Compensation | |||||||||||||||||||||||||
| Vininder Singh | 2025 | $ | 400,000 | $ | - | $ | 288,637 | $ | 129,000 | $ | 347 | [2] | $ | - | $ | - | $ | 817,984 | ||||||||||||||||
| Chief Executive Officer and Director | 2024 | $ | 400,000 | $ | - | $ | - | $ | 230,680 | $ | - | $ | - | $ | - | $ | 630,680 | |||||||||||||||||
| Josh Blacher | 2025 | $ | 235,069 | $ | - | $ | 19,051 | $ | - | $ | - | $ | - | $ | - | $ | 254,120 | |||||||||||||||||
| Chief Financial Officer | 2024 | $ | 10,631 | $ | - | $ | - | $ | - | $ | - | $ | - | $ | - | $ | 10,631 | |||||||||||||||||
[1] Represents annual value of stock grants and RSU grants issued during fiscal year 2025 under our 2022 Equity Incentive Plan.
[2] Comprised of amounts relating to employer-sponsored group term life insurance and benefit provided by interest-free loan.
Employment Agreements
On May 16, 2022, we entered into an employment agreement with Vininder Singh, pursuant to which he received an annual base salary of $400,000, which is subject to bi-annual review by the Company. Mr. Singh is also eligible for an annual bonus based on the achievement of certain goals and performance criteria established by the Board. Mr. Singh’s target annual bonus for the fiscal years ended 2022 through 2025 was a minimum of twenty (20%) percent of the current base salary, with a maximum payout of up to one-hundred (100%) percent based on target achievement. Mr. Singh will also be eligible to participate in the Company’s stock incentive plan, subject to Board approval. The agreement with Mr. Singh shall continue until either his resignation, termination for cause by the Company, or death or disability of Mr. Singh.
Consulting Agreements
On December 12, 2024, the Board of Directors appointed Josh Blacher to serve as the Company’s Chief Financial Officer. In such capacity, Mr. Blacher will serve as the principal financial officer and principal accounting officer of the Company. Mr. Blacher provides services to the Company as an independent contractor pursuant to a master services agreement entered into on December 13, 2024 (the “Consulting Agreement”) by the Company and Danforth Advisors, LLC (“Danforth”). Pursuant to the Consulting Agreement, we will pay Danforth cash compensation at a rate of $525 per hour for Mr. Blacher’s services, which is subject to an optional increase by Danforth of up to 5% on January 1 of each year. Mr. Blacher will receive no compensation directly from the Company. As of the date of this filing, no increase to the hourly rate has been implemented in 2026 pursuant to this provision.
| 50 |
Director Compensation
The following table summarizes the compensation paid to our executive and non-executive directors during the year ended December 31, 2025.
| Name | Fees Earned or Paid in Cash[1] | Stock Awards[2] | Option Awards[3] | All Other Compensation | Nonequity Incentive Plan Compensation | Nonqualified Deferred Compensation Earnings | Total Compensation | |||||||||||||||||||||
| Vininder Singh[4] | $ | - | $ | - | $ | - | $ | - | $ | - | $ | - | $ | - | ||||||||||||||
| R. Donald Elsey | $ | 45,000 | $ | 10,824 | $ | 16,500 | $ | - | $ | - | $ | - | $ | 72,324 | ||||||||||||||
| William Enright | $ | 45,000 | $ | 10,824 | $ | 16,500 | $ | - | $ | - | $ | - | $ | 72,324 | ||||||||||||||
| Jason D. Hanson | $ | 45,000 | $ | 10,824 | $ | 16,500 | $ | - | $ | - | $ | - | $ | 72,324 | ||||||||||||||
[1] Represents cash compensation for service as a director and as chair of a board committee during the fiscal year 2025.
[2] Represents annual value of stock grants and RSU grants issued during fiscal year 2025 under our 2022 Equity Incentive Plan.
[3] Represents annual value of stock options issued during fiscal year 2025 under our 2022 Equity Incentive Plan.
[4] Mr. Singh did not receive additional compensation for his service as a director of our Company during the fiscal year 2025.
Pension, Retirement or Similar Benefit Plans
There are no arrangements or plans in which we provide pension, retirement or similar benefits for directors or executive officers. We have no material bonus or profit-sharing plans pursuant to which cash or non-cash compensation is or may be paid to our directors or executive officers, except that equity awards may be granted at the discretion of the Board or a committee thereof.
Indebtedness of Directors, Senior Officers, Executive Officers and Other Management
None of our directors, executive officers or any associate or affiliate of our Company during the last two fiscal years is or has been indebted to our Company by way of guarantee, support agreement, letter of credit or other similar agreement or understanding currently outstanding.
Equity Compensation Plans
In November 2022, our Board of Directors and shareholders adopted the 2022 Equity Incentive Plan (the “Plan”). Pursuant to the Plan, we are authorized to grant options and other equity awards to officers, directors, employees and consultants. The exercise price of each share of common stock purchasable under an award issued pursuant to the Plan shall be determined by our compensation committee, in its sole discretion, at the time of grant, but shall not be less than 100% of the fair market value of such share of common stock on the date the award is granted, subject to adjustment and conditions further described in the Plan. Our compensation committee shall also have sole authority to set the terms of all awards at the time of grant. In October 2025, the Company’s stockholders approved an amendment to increase shares available for grant under the Plan by 750,000. As of December 31, 2025, there are 891,975 shares available under the Plan.
| 51 |
Outstanding Equity Awards at Fiscal Year-End
The following table summarizes the outstanding equity awards held by each named executive officer as of December 31, 2025. This table includes unexercised and unvested options and equity awards.
| Outstanding Equity Awards as of December 31, 2025 | ||||||||||||||||||||||||||||||||||||||
| Option Awards | Stock Awards | |||||||||||||||||||||||||||||||||||||
| Name | Date of Grant | Number of securities underlying unexercised options (#) exercisable | Number of securities underlying unexercised options (#) unexerciseable (1) | Equity incentive plan awards: Number of securities underlying unexercised unearned options (#) | Option exercise price ($) | Option expiration date | Number of shares of stock that have not vested (#) | Market value of shares of stock that have not vested ($) (2) | Equity incentive plan awards: number of unearned shares, units, or other rights that have not vested (#) | Equity incentive plan awards: market or payout value of unearned shares, units or other rights that have not vested ($) (2) | ||||||||||||||||||||||||||||
| Vininder Singh | 1/18/2024 | 52,930 | 26,070 | - | $ | 3.89 | 1/18/2034 | - | - | - | - | |||||||||||||||||||||||||||
| Vininder Singh | 1/28/2025 | 25,000 | 50,000 | - | $ | 2.26 | 1/28/2035 | - | - | - | - | |||||||||||||||||||||||||||
| Vininder Singh | 8/22/2025 | - | - | - | - | - | 152,273 | $ | 134,427 | - | - | |||||||||||||||||||||||||||
| Josh Blacher | 8/22/2025 | - | - | - | - | - | 10,050 | $ | 8,872 | - | - | |||||||||||||||||||||||||||
| (1) | The standard vesting schedule for all stock option grants is vesting over two years with one-third vesting on the date of grant, one-third vesting on the first anniversary of the date of grant, and one-third vesting on the second anniversary of the date of grant. | |
| (2) | Calculated based on the closing price of the common stock on the Nasdaq Capital Market on December 31, 2025 of $0.8828 per share. |
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
The following table sets forth certain information regarding the beneficial ownership of our common stock as of March 1, 2026 by:
| ● | each of our named executive officers; | |
| ● | each of our directors; | |
| ● | all of our current directors and executive officers as a group; and | |
| ● | each stockholder known by us to own beneficially more than 5% of our common stock. |
Beneficial ownership is determined in accordance with the rules of the SEC and includes voting or investment power with respect to the securities. Shares of common stock that may be acquired by an individual or group within 60 days of March 1, 2026, pursuant to the exercise of options or warrants, vesting of common stock or conversion of convertible debt, are deemed to be outstanding for the purpose of computing the percentage ownership of such individual or group, but are not deemed to be outstanding for the purpose of computing the percentage ownership of any other person shown in the table. Percentage of ownership is based on 12,664,387 shares of common stock issued and outstanding as of March 1, 2026.
| 52 |
Except as otherwise indicated, all shares are owned directly. Unless otherwise indicated, the address of each of the persons shown is c/o BullFrog AI Holdings, Inc., 325 Ellington Blvd., Unit 317, Gaithersburg, MD 20878.
| Name of Beneficial Owner | Common Stock Beneficially Owned | Percentage of Common Stock | ||||||
| Directors and Officers: | ||||||||
| Vininder Singh | ||||||||
| Chief Executive Officer and Director (1) | 2,496,446 | 19.51 | % | |||||
| Josh Blacher | ||||||||
| Chief Financial Officer (2) | 4,950 | * | ||||||
| R. Donald Elsey (3) | 57,813 | * | ||||||
| William Enright (4) | 62,813 | * | ||||||
| Jason D. Hanson (3) | 57,813 | * | ||||||
| All officers and directors as a group (6 persons) | 2,679,835 | 20.68 | % | |||||
| Beneficial owners of more than 5% | ||||||||
| Tivoli Trust (5) | 904,391 | 6.69 | % | |||||
* Less than 1%
| (1) | Comprised of 2,367,446 shares of common stock and 129,000 stock options that have vested as of March 1, 2026 or will vest within 60 days of such date. |
| (2) | Comprised of 4,950 shares of common stock. |
| (3) | Comprised of 2,813 shares of common stock and 55,000 stock options that have vested as of March 1, 2026 or will vest within 60 days of such date. |
| (4) | Comprised of 7,813 shares of common stock and 55,000 stock options that have vested as of March 1, 2026 or will vest within 60 days of such date. |
| (5) | Comprised of 73,449 shares of non-voting Series A Preferred Stock, 115,185 warrants exercisable at $2.50 per share and 54,714 shares of Common Stock. The common stock total assumes the conversion of all Series A Preferred Stock into common stock in an amount equal to ten shares of common stock for each one share of Series A Preferred Stock. |
Securities Authorized for Issuance under Equity Compensation Plans
General. In November 2022, our Board of Directors adopted our 2022 Equity Incentive Plan (the “2022 Plan”) and the 2022 Plan was submitted to our stockholders for approval. Our 2022 Plan became effective immediately on adoption. Our 2022 Plan replaces our previous incentive plan. However, awards outstanding under our previous incentive plan will continue to be governed by their existing terms.
Share Reserve. The number of shares of our common stock originally available for issuance under our 2022 Plan was 900,000 shares. Notwithstanding the number of shares available for issuance, on the first day of each year commencing January 1, 2023, or the first business day of the calendar year if the first day of the calendar year falls on a Saturday or Sunday, the number of shares eligible for awards under the 2022 Plan will automatically increase to an amount equal to 15% of the total number of shares of common stock outstanding as of December 31st of the preceding fiscal year. Accordingly, as of January 1, 2026, there were 1,237,732 shares available for issuance under the 2022 Plan.
The following table shows, as of December 31, 2025, the Company’s equity compensation plans under which the Company’s equity securities are authorized for issuance:
| Plan Category | Number of securities to be issued upon exercise of outstanding options, warrants and rights | Weighted-average exercise price of outstanding options, warrants and rights (2) | Number of securities remaining available for future issuance under equity compensation plans | |||||||||
| Equity compensation plans approved by security holders | 1,056,530 | (1) | $ | 3.53 | 891,975 | |||||||
| Equity compensation plans not approved by security holders | 693,176 | $ | 1.59 | 0 | ||||||||
| Total | 1,749,706 | $ | 2.58 | 891,975 | ||||||||
| (1) | Includes 342,030 outstanding restricted stock units for which there is no exercise price. | |
| (2) | Includes the weighted-average exercise price of stock options and warrants only. |
| 53 |
Policies and Practices Related to the Grant of Certain Equity Awards
We grant equity awards to our executives, including stock options, as part of their total compensation. In 2025, we did not grant stock options on any date that, in relation to our disclosure of material nonpublic information, would require us to provide the tabular disclosures of Item 402(x)(2) of Regulation S-K. Accordingly, we have no specific policy or practice on the timing of awards of such options in relation to the disclosure of material nonpublic information by us. In the event that we determine to grant new awards of such options, the Compensation Committee will evaluate the appropriate steps to take in relation to the foregoing. Our insider trading policy also provides guidelines around the repurchases of our securities, which would generally only made pursuant to a Rule 10b5-1 trading plan established when we are not in possession of material nonpublic information.
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
Other than with respect to compensation arrangements, including employment and indemnification arrangements discussed in this Annual Report on Form 10-K, there have been no transactions since January 1, 2024, in which the amount involved in the transaction exceeded or will exceed the lesser of $120,000 or one percent of the average of our total assets as at year-end for the last two completed fiscal years, and to which any of our directors, executive officers or beneficial holders of more than 5% of our capital stock, or any immediate family member of, or person sharing the household with, any of these individuals, had or will have a direct or indirect material interest.
In October 2022, we entered into an exchange agreement with an investor whereby all 734,493 shares of his common stock were exchanged into 73,449 shares of Series A Convertible Preferred Stock that converts to common stock at a rate of 10 shares of common stock for one share of preferred stock. The Series A Preferred Stock is the economic equivalent of the common stock but has no voting rights and is subject to a blocker which prohibits the conversion into common stock if it would result in the investor owning more than 4.99% of the Company’s outstanding common stock at such time.
Policies and Procedures for Related Party Transactions
For purposes of our policy only, a related person transaction is a transaction, arrangement or relationship, or any series of similar transactions, arrangements, or relationships, in which we and any related person are, were or will be participants in which the amount involved exceeds the lesser of $120,000 or 1% of the average of our total assets at year-end. Transactions involving compensation for services provided to us as an employee or director are not covered by this policy. A related person is any executive officer, director, or beneficial owner of more than 5% of any class of our voting securities, including any of their immediate family members and any entity owned or controlled by such persons.
Under the policy, if a transaction has been identified as a related person transaction, including any transaction that was not a related person transaction when originally consummated or any transaction that was not initially identified as a related person transaction prior to consummation, our management must present information regarding the related person transaction to our audit committee, or, if audit committee approval would be inappropriate, to another independent body of our Board of Directors, for review, consideration and approval or ratification. The presentation must include a description of, among other things, the material facts, the interests, direct and indirect, of the related persons, the benefits to us of the transaction and whether the transaction is on terms that are comparable to the terms available to or from, as the case may be, an unrelated third party or to or from employees generally. Under the policy, we will collect information that we deem reasonably necessary from each director, executive officer and, to the extent feasible, significant stockholder to enable us to identify any existing or potential related-person transactions and to effectuate the terms of the policy. In addition, under our code of business conduct and ethics, our employees and directors will have an affirmative responsibility to disclose any transaction or relationship that reasonably could be expected to give rise to a conflict of interest. In considering related person transactions, our audit committee, or other independent body of our Board of Directors, will take into account the relevant available facts and circumstances including, but not limited to:
| ● | the risks, costs and benefits to us; |
| 54 |
| ● | the impact on a director’s independence in the event that the related person is a director, immediate family member of a director or an entity with which a director is affiliated; |
| ● | the availability of other sources for comparable services or products; and |
| ● | the terms available to or from, as the case may be, unrelated third parties or to or from employees generally. |
The policy requires that, in determining whether to approve, ratify or reject a related person transaction, our audit committee, or other independent body of our Board of Directors, must consider, in light of known circumstances, whether the transaction is in, or is not inconsistent with, our best interests and those of our stockholders, as our audit committee, or other independent body of our Board of Directors, determines in the good faith exercise of its discretion.
ITEM 14. PRINCIPAL ACCOUNTING FEES AND SERVICES
The following table summarizes the fees billed by M&K CPAs for the fiscal years ended December 31, 2025 and 2024, inclusive of out-of-pocket expenses.
Pre-Approval Policy
Our audit committee was formed upon the consummation of our initial public offering. As a result, the audit committee did not pre-approve all of the foregoing services, although any services rendered prior to the formation of our audit committee were approved by our board of directors. Since the formation of our audit committee, and on a going-forward basis, the audit committee has and will pre-approve all auditing services and permitted non-audit services to be performed for us by our auditors, including the fees and terms thereof (subject to the de minimis exceptions for non-audit services described in the Exchange Act which are approved by the audit committee prior to the completion of the audit).
| Year Ended December 31, | ||||||||
| Fee Category | 2025 | 2024 | ||||||
| Audit fees(1) | $ | 82,250 | $ | 44,725 | ||||
| Audit-related fees(2) | 25,250 | 26,820 | ||||||
| Tax fees(3) | - | - | ||||||
| All other fees(4) | - | - | ||||||
| Total fees | $ | 107,500 | $ | 71,545 | ||||
| (1) | Audit fees consist of fees for professional services rendered in connection with the annual audit of our consolidated financial statements, the review of our quarterly condensed consolidated financial statements and consultations on accounting matters directly related to the audit. |
| (2) | Audit-related fees consist of fees for professional services rendered in connection with submissions of Registration Statements on Form S-1 or Form S-3 for our initial public offering and subsequent financings. |
| (3) | Tax fees consist of fees for professional services for tax compliance, tax advice and tax planning. |
| (4) | All other fees consist of fees related to engagement administration. |
| 55 |
PART IV
Item 15. Exhibits, Financial Statement Schedules
| a) | Financial Statements |
For a list of the consolidated financial statements included herein, see Index to Consolidated Financial Statements on page F-1 of this Annual Report, which is incorporated into this Item by reference.
| b) | Exhibits |
| 56 |
| 57 |
| ● | Filed herewith. |
| ** | Certain portions of the exhibit have been omitted in accordance with Regulation S-K Item 601 (b)(10). The Registrant agrees to furnish supplementally an unredacted copy of the exhibit to the SEC upon its request. |
ITEM 16. FORM 10-K SUMMARY
None.
| 58 |
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| March 19, 2026 | BullFrog AI Holdings, Inc. | |
| By: | /s/ Vininder Singh | |
| Vininder Singh | ||
| Chief Executive Officer and Director (Principal Executive Officer) | ||
| By: | /s/ Josh Blacher | |
| Josh Blacher | ||
| Chief Financial Officer (Principal Financial and Accounting Officer) | ||
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| Signature | Title | Date | |||
| By: | /s/ Vininder Singh | Chief Executive Officer and Chairman (Principal Executive Officer) |
March 19, 2026 | ||
| Vininder Singh | |||||
| By: | /s/ Josh Blacher | Chief Financial Officer (Principal Financial and Accounting Officer) |
March 19, 2026 | ||
| Josh Blacher | |||||
| By: | /s/ R. Donald Elsey | Director | March 19, 2026 | ||
| R. Donald Elsey | |||||
| By: | /s/ William Enright | Director | March 19, 2026 | ||
| William Enright | |||||
| By: | /s/ Jason D. Hanson | Director | March 19, 2026 | ||
| Jason D. Hanson | |||||
| 59 |
BULLFROG AI HOLDINGS, INC.
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
| F-1 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders of BullFrog AI Holdings, Inc.
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated balance sheets of BullFrog AI Holdings, Inc. (the “Company”) as of December 31, 2025 and 2024, and the related consolidated statements of operations, changes in stockholders’ equity, and cash flows for the years ended December 31, 2025 and 2024, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2025 and 2024 and the results of its operations and its cash flows for the two-year period ended December 31, 2025, in conformity with accounting principles generally accepted in the United States of America.
Going Concern
The accompanying consolidated financial statements have been prepared assuming the Company will continue as a going concern. As discussed in Note 1 to the consolidated financial statements, the Company has incurred recurring losses from operations and has not yet achieved profitable operations as of December 31, 2025 which raises substantial doubt about its ability to continue as a going concern. Management’s plans regarding these matters are also described in Note 1. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matter
The critical audit matter communicated below is a matter arising from the current period audit of the consolidated financial statements that were communicated or required to be communicated to the audit committee and that: (1) relate to accounts or disclosures that are material to the consolidated financial statements and (2) involved our especially challenging, subjective, or complex judgments. The communication of a critical audit matter does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matter below, providing separate opinions on the critical audit matter or on the accounts or disclosures to which they relate.
Revenue Recognition
Auditing the Company’s revenue recognition required significant judgment in applying ASC 606. Specifically, auditing management’s evaluation of the agreement with a customer involved assessing the identification and allocation of standalone transaction prices to performance obligations under ASC 606. As such, revenue recognition was identified as a Critical Audit Matter, as improper revenue recognition could materially misstate the financial statements and key financial indicators.
To address these risks, we reviewed and assessed a customer agreement and management’s evaluation of key terms and related disclosures. We also performed substantive audit procedures to test the appropriateness, accuracy, and completeness of recorded revenue transactions.
| /s/
|
|
| We have served as the Company’s auditor since 2021. | |
| March 19, 2026 |
| F-2 |
BULLFROG AI HOLDINGS, INC.
CONSOLIDATED BALANCE SHEETS
| December 31, | ||||||||
| 2025 | 2024 | |||||||
| Assets | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | $ | ||||||
| Prepaid expenses and other assets | ||||||||
| Total current assets | ||||||||
| Restricted cash | ||||||||
| Property and equipment, net | ||||||||
| Investments | ||||||||
| Total assets | $ | $ | ||||||
| Liabilities and Stockholders’ Equity | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | $ | ||||||
| Accrued expenses | ||||||||
| Total current liabilities | ||||||||
| Total liabilities | ||||||||
| Stockholders’ equity: | ||||||||
| Series A Convertible Preferred stock, $ par value, shares authorized; shares issued and outstanding as of December 31, 2025 and 2024. | ||||||||
| Common stock, $ par value, shares authorized; and shares issued and outstanding as of December 31, 2025 and 2024, respectively. | ||||||||
| Additional paid-in capital | ||||||||
| Accumulated deficit | ( | ) | ( | ) | ||||
| Total stockholders’ equity | ||||||||
| Total liabilities and stockholders’ equity | $ | $ | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-3 |
BULLFROG AI HOLDINGS, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
| Year Ended December 31, | ||||||||
| 2025 | 2024 | |||||||
| Revenue: | ||||||||
| Collaboration revenue | $ | $ | ||||||
| Total revenue | ||||||||
| Cost of revenue: | ||||||||
| Cost of collaboration revenue | ||||||||
| Total cost of revenue | ||||||||
| Gross profit | ||||||||
| Operating expenses: | ||||||||
| Research and development | ||||||||
| General and administrative | ||||||||
| Total operating expenses | ||||||||
| Loss from operations | ( | ) | ( | ) | ||||
| Other income (expense), net | ||||||||
| Interest expense | ( | ) | ( | ) | ||||
| Interest income | ||||||||
| Total other income (expense), net | ||||||||
| Net loss | ( | ) | ( | ) | ||||
| Deemed dividend related to warrant exercise price adjustment | ( | ) | ||||||
| Net loss attributable to common stockholders | $ | ( | ) | $ | ( | ) | ||
| Net loss per common share attributable to common stockholders - basic and diluted | $ | ) | $ | ) | ||||
| Weighted average number of shares outstanding - basic and diluted | ||||||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-4 |
BULLFROG AI HOLDINGS, INC.
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ EQUITY
Series A Preferred Stock | Common Stock | Additional Paid-in | Accumulated | Total Stockholders’ | ||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Capital | Deficit | Equity | ||||||||||||||||||||||
| Balance at December 31, 2023 | $ | | $ | $ | | $ | ( | ) | $ | | ||||||||||||||||||
| Stock-based compensation | - | - | ||||||||||||||||||||||||||
| Issuance of common stock and warrants, net of issuance costs | - | |||||||||||||||||||||||||||
| Issuance of common stock pursuant to warrant exercises | - | |||||||||||||||||||||||||||
| Deemed dividend related to warrant price adjustment | - | - | ( | ) | ||||||||||||||||||||||||
| Net loss | - | - | ( | ) | ( | ) | ||||||||||||||||||||||
| Balance at December 31, 2024 | ( | ) | ||||||||||||||||||||||||||
| Stock-based compensation | - | |||||||||||||||||||||||||||
| Issuance of common stock pursuant to warrant exercises | - | ( | ) | |||||||||||||||||||||||||
| Issuance of common stock, net of issuance costs | - | |||||||||||||||||||||||||||
| Issuance of common stock as commitment fee in advance of equity transaction | - | |||||||||||||||||||||||||||
| Net loss | - | - | ( | ) | ( | ) | ||||||||||||||||||||||
| Balance at December 31, 2025 | $ | $ | $ | $ | ( | ) | $ | |||||||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-5 |
BULLFROG AI HOLDINGS, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
| Year Ended December 31, | ||||||||
| 2025 | 2024 | |||||||
| Cash flows from operating activities: | ||||||||
| Net loss | $ | ( | ) | $ | ( | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
| Depreciation | ||||||||
| Stock-based compensation | ||||||||
| Non-cash consideration received pursuant to revenue agreements | ( | ) | ||||||
| Changes in operating assets and liabilities: | ||||||||
| Prepaid expenses and other assets | ||||||||
| Accounts payable | ( | ) | ||||||
| Accrued expenses | ||||||||
| Net cash used in operating activities | ( | ) | ( | ) | ||||
| Cash flows from investing activities: | ||||||||
| Net cash used in investing activities | ||||||||
| Cash flows from financing activities: | ||||||||
| Proceeds from sale of common stock from ATM, net of issuance costs | ||||||||
| Proceeds from issuance of common stock and warrants, net of issuance costs | ||||||||
| Proceeds from warrant exercises | ||||||||
| Issuance costs paid in advance of equity financing | ( | ) | ||||||
| Proceeds from short term insurance financing | ||||||||
| Payments on short term insurance financing | ( | ) | ( | ) | ||||
| Net cash provided by financing activities | ||||||||
| Net (decrease) increase in cash and cash equivalents | ( | ) | ||||||
| Cash and cash equivalents and restricted cash, beginning of period | ||||||||
| Cash and cash equivalents and restricted cash, end of period | $ | $ | ||||||
| Supplemental cash flow information: | ||||||||
| Cash paid for interest | $ | $ | ||||||
| Cash paid for taxes | ||||||||
| Supplemental non-cash activity | ||||||||
| Issuance of common stock as commitment fee in advance of equity transaction | $ | $ | ||||||
The accompanying notes are an integral part of these consolidated financial statements.
| F-6 |
BULLFROG AI HOLDINGS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
December 31, 2025 and 2024
1. Organization and Nature of Business
Description of Business
BullFrog AI Holdings, Inc. (“we”, “our” or the “Company”) was incorporated in the State of Nevada in February 2020. BullFrog AI Holdings, Inc. is the parent company of BullFrog AI, Inc. and BullFrog AI Management, LLC, which were incorporated in Delaware and Maryland, in 2017 and 2021, respectively. All the Company’s operations are currently conducted through BullFrog AI Holdings, Inc., which began operations in February 2020. The Company is focused specifically on advanced artificial intelligence and machine learning (“AI/ML”) driven analysis of complex data sets in medicine and healthcare. The Company’s objective is to utilize its AI/ML platform to provide a precision medicine approach to drug asset enablement through external partnerships and selective internal development.
Most new therapeutics will fail at some point in preclinical or clinical development. These failures are the primary drivers for the high cost of developing new therapeutics. A major challenge in developing new therapeutics is efficiently integrating the complex, high-dimensional data generated at each stage of development to help de-risk subsequent stages of the process. AI/ML have emerged as a digital solution to help address this problem.
The Company uses AI/ML to advance medicines for both internal and external projects. Currently, most AI/ML platforms still fall short in their ability to synthesize disparate, high-dimensional data for actionable insight. The Company’s analytical platform is composed of an ensemble of state-of-the-art machine learning and artificial intelligence models. The Company’s core platform technology, bfLEAP™, is an analytical AI/ML platform developed at The Johns Hopkins University Applied Physics Laboratory (“JHU-APL”), which the Company believes is able to surmount the challenges of scalability and flexibility currently hindering researchers and clinicians by providing a more precise, multi-dimensional understanding of their data. The Company is deploying its analytical platform, including bfLEAP™, in several critical stages of development of internal programs and to customers through strategic partnerships and collaborations with the intention of streamlining data analytics in therapeutics development, decreasing the overall development costs by decreasing failure rates for new therapeutics, and impacting the lives of countless patients that may otherwise not receive the therapies they need.
The proprietary analytical platform utilizes both supervised and unsupervised machine learning. As such, it can reveal real and meaningful connections in the data without the need for a priori hypothesis. Algorithms used in the platform are designed to handle highly imbalanced data sets and successfully identify combinations of factors that are associated with outcomes of interest. The Company’s platform leverages models that use both correlative and causative machine learning and artificial intelligence approaches which provide a comprehensive approach to predictive analysis that is expected to lead to meaningful insights including the molecular drivers of disease. In this regard, the Company continues to advance its internal target discovery initiatives through access to proprietary datasets, including those available under its strategic data and commercialization agreements with the Lieber Institute for Brain Development (“LIBD”).
The Company’s goal is to improve the odds of success at all stages of pre-clinical and clinical development for in-house programs and for its strategic partners, collaborators, and customers. The Company’s business model includes supporting clinical trial optimization efforts for ongoing studies and rescuing late stage failed drugs (i.e., Phase II or Phase III clinical trial failures) by bringing them in-house for development prior to eventual divestiture; although, the Company also considers entering collaborations for earlier stage drugs. The Company pursues its drug asset enhancement business by leveraging the powerful and proven bfLEAP™ AI/ML platform initially developed at JHU-APL. The Company believes the bfLEAP™ analytics platform is a potentially disruptive tool for analysis of pre-clinical and clinical data sets, such as the robust pre-clinical and clinical trial datasets being generated in translational R&D and clinical trial settings.
Liquidity and Going Concern
The
Company has generated negative cash flows from operations and operated at a net loss since inception. As of December 31, 2025, the Company
has a cash balance of approximately $
| F-7 |
Accordingly, the Company will require additional capital to continue to execute its strategy. The Company anticipates securing this additional capital through various avenues including revenues from services, licensing agreements and collaborative arrangements within its operating business and/or the selling of equity securities or entry into debt transactions. Although management believes that such funding sources will be available, including pursuant to the Company’s at-the-market common stock sales facility provided by our ATM Agreement and pursuant to the Company’s equity line of credit facility provided by our purchase agreement with Lincoln Park Capital Fund, LLC, there can be no assurance that any such arrangements will provide sufficient capital when needed to allow the Company to continue its operations, or if available, be on terms acceptable to it. If the Company does not raise sufficient funds in a timely manner, among other things, it may be forced to delay, scale back or eliminate some or all of its research and product development programs and capital expenditures or enter into arrangements on unfavorable terms. The Company does not currently have commitments for future funding from any source other than those noted above. Furthermore, the issuance of additional equity securities may be significantly dilutive to the Company’s current shareholders.
On
August 21, 2025, the Company received a letter from the listing staff of The Nasdaq Stock Market LLC (“Nasdaq”) that the
Company was no longer in compliance with the minimum stockholders’ equity requirement for continued listing on Nasdaq pursuant
to Nasdaq Listing Rule 5550(b)(1) (the “Stockholders’ Equity Rule”). The Stockholders’ Equity Rule requires
companies listed on the Nasdaq Capital Market to maintain stockholders’ equity of at least $
On February 10, 2026, the Company received a letter from Nasdaq notifying the Company that, for the last 30 consecutive business days, the closing bid price for the Company’s common stock, par value $ per share (the “Common Stock”), was below $ per share, which is the minimum closing bid price required for continued listing on the Nasdaq Global Market (the “Minimum Bid Price Requirement”) pursuant to Nasdaq Listing Rule 5550(a)(2) (the “Bid Price Notice”). The Bid Price Notice had no immediate effect on the listing of the Company’s Common Stock and tradeable warrants. As such, the Company’s Common Stock will continue to trade on the Nasdaq Capital Market under the symbol “BFRG,” and its tradeable warrants will continue to trade on the Nasdaq Capital Market under the symbol “BFRGW.” In accordance with Nasdaq Listing Rule 5810(c)(3)(A), the Company is provided a compliance period of 180 calendar days from the date of the Bid Price Notice, or until August 10, 2026, to regain compliance with the Minimum Bid Price Requirement. If at any time during the 180-calendar day grace period, the closing bid price of the Company’s Common Stock is at least $ per share for a minimum of ten consecutive business days (unless the Nasdaq staff exercises its discretion to extend this ten business day period pursuant to Nasdaq Listing Rule 5810(c)(3)(H)), Nasdaq will provide the Company written confirmation of compliance, and the matter will be closed. If the Company does not regain compliance during the initial 180-calendar day compliance period, the Company may be provided a second 180-calendar day period to regain compliance. If the Company does not regain compliance within the allotted compliance periods, including any extensions that may be granted by Nasdaq, the Company’s listed securities will be subject to delisting. The Company would thereafter have the right to appeal a determination to delist the Company’s securities, and the Company’s securities would remain listed on the Nasdaq Capital Market until the completion of the appeal process. The Company intends to monitor the closing bid price of its Common Stock and assess potential options to regain compliance with Nasdaq’s Listing Rules. While the Company plans to review all available options, there can be no assurance that the Company will regain compliance with the Minimum Bid Price Requirement during the compliance period, secure a second 180-day period to regain compliance with the Minimum Bid Price Requirement, or maintain compliance with the other Nasdaq listing requirements.
| F-8 |
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern, which contemplates continuity of operations, realization of assets, and satisfaction of liabilities in the ordinary course of business. Accordingly, these consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
2. Summary of Significant Accounting Policies
Basis of Presentation
The accompanying consolidated financial statements include the accounts of BullFrog AI Holdings, Inc. and its wholly owned subsidiaries and have been prepared in conformity with United States generally accepted accounting principles (“GAAP”). All intercompany accounts and transactions have been eliminated in consolidation.
Use of Estimates
The preparation of consolidated financial statements in conformity with GAAP requires the Company to make estimates and assumptions that affect the amounts reported in the consolidated financial statements and accompanying notes. These estimates include, but are not limited to, revenue recognition, stock-based compensation, allowances for doubtful accounts, recoverability of deferred tax assets, valuation of the Company’s equity investment, and certain other accrued liabilities. Actual results could differ from these estimates.
Segment Reporting
The Company’s chief operating decision maker (“CODM”) is the Company’s Chief Executive Officer. The CODM is assisted in his responsibilities of making decisions regarding resource allocation and performance assessment by the leadership team, consisting of executives and vice presidents.
The Company views its operations and manages its business as one operating segment, focused on advancing drug development using AI/ML to analyze complex data sets in medicine and healthcare. Segment profit or loss is measured as the Company’s net income (loss) as reported on the Company’s Statement of Operations. The Company monitors its cash and cash equivalents, as reported on the Company’s Balance Sheets, to determine funding for its research and development.
The CODM assesses Company performance through the achievement of revenue, cost optimization, and target identification goals. In addition to the Company’s Statement of Operations, the CODM is regularly provided with budgeted and forecasted expense information which is used to determine the Company’s liquidity needs and cash allocation.
Revenue Recognition
The Company recognizes revenue based on the following five step model:
| ● | Identification of the contract with a customer |
This step outlines the criteria that must be met when establishing a contract with a customer to supply goods or services.
| ● | Identification of the performance obligations in the contract |
This step describes how distinct performance obligations in the contract must be handled.
| ● | Determination of the transaction price |
This step outlines what must be considered when establishing the transaction price, which is the amount the business expects to receive for transferring the goods and services to the customer.
| ● | Allocation of the transaction price to the performance obligations in the contract |
This step outlines guidelines for allocating the transaction price across the contract’s separate performance obligations, and is what the customer agrees to pay for the goods and services.
| ● | Recognition of revenue when, or as, the Company satisfies a performance obligation |
Revenue can be recognized as the business meets each performance obligation. This step specifies how that should happen.
| F-9 |
Contract Services
The Company anticipates that the majority of its revenues that may be recognized will result from discovery and monetization of new drug targets and intellectual property from data use partnerships focused on analysis of rich proprietary datasets. The target market for monetization will primarily be mid-size to large biopharmaceutical organizations seeking to build their new drug target pipeline through collaboration agreements with companies such as BullFrog. A secondary revenue channel is fee-for-service partnerships and collaborations with biopharmaceutical companies and other organizations of all sizes that have challenges analyzing data throughout the drug development process. The Company provides customers with an analysis of large complex data sets using the Company’s proprietary AI/ML platform. This platform is aimed at predicting targets of interest, patterns, relationships, anomalies, and molecular drivers of disease. The Company believes that there will be additional on-going work requested from partners; therefore, the service model utilizes a master services agreement with work or task orders issued for discrete analysis performed at the discovery, preclinical, or clinical stages of drug development. The Company will receive fees related to such agreements in either cash, the equity of its partners, or other consideration and, in some instances, the potential for rights to new intellectual property generated from the analysis. Once data analysis and the analysis report are complete, the Company delivers the analysis set to the customer and recognizes revenue at that point in time.
Investments
The Company currently has a single investment in equity securities issued by a privately held entity. The Company entered into a strategic collaboration agreement and received such equity securities as remuneration for services rendered. The Company has elected to account for this investment using the measurement alternative as the investment does not have a readily determinable fair value. Pursuant to this alternative, the investment will be carried at its estimated fair value calculated as its cost minus any impairment. The Company will adjust the investment to fair value only when it identifies observable price changes in orderly transactions for identical or similar investments of the same issuer. The Company will evaluate the investment at each reporting period to determine whether the investment is impaired.
Financial Instruments
The carrying value of short-term instruments, including cash and cash equivalents, accounts payable and accrued expenses approximate fair value due to the relatively short period to maturity for these instruments. The Company has elected to account for its single investment using the measurement alternative and it is considered a financial instrument accounted for at fair value on a non-recurring basis. Fair value is defined as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. Valuation techniques used to measure fair value maximize the use of observable inputs and minimize the use of unobservable inputs. The Company utilizes a three-level valuation hierarchy for disclosures of fair value measurements, defined as follows:
Level 1 - inputs to the valuation methodology are quoted prices (unadjusted) for identical assets or liabilities in active markets.
Level 2 - inputs to the valuation methodology include quoted prices for similar assets and liabilities in active markets, and inputs that are observable for the assets or liabilities, either directly or indirectly, for substantially the full term of the financial instruments.
Level 3 - inputs to the valuation methodology are unobservable and significant to the fair value.
The Company does not have any assets or liabilities that are required to be measured and recorded at fair value on a recurring basis.
Cash and Cash Equivalents, Restricted Cash, and Concentration of Credit Risk
The Company considers cash to consist of cash on hand and temporary investments having an original maturity of 90 days or less that are readily convertible into cash.
Restricted cash represents cash balances that are legally or contractually restricted as to withdrawal or usage. These amounts are not available for general operating purposes and are maintained in separate accounts or otherwise designated for specific uses.
The Company’s financial instruments that are exposed to a concentration of credit risk are cash and accounts receivable. Occasionally, the Company’s cash in interest-bearing accounts may exceed FDIC insurance limits. The financial stability of these institutions is periodically reviewed by senior management.
| F-10 |
The following table provides a reconciliation of cash and cash equivalents and restricted cash reported within the consolidated balance sheets that sum to the total of the same amounts shown in the consolidated statements of cash flows:
| December 31, | ||||||||
| 2025 | 2024 | |||||||
| Cash and cash equivalents | $ | $ | ||||||
| Restricted cash | ||||||||
| Total cash and cash equivalents and restricted cash in the consolidated statements of cash flows | $ | $ | ||||||
Cost of Revenue
Cost of revenue consists primarily of the allocation of personnel costs (e.g. payroll, benefits, and consulting fees) of our employees and third-party consultants directly attributable to the satisfaction of our performance obligations under our revenue arrangements.
Property and Equipment, net
Property and equipment are stated at cost. When retired or otherwise disposed, the related carrying value and accumulated depreciation are removed from the respective accounts and the net difference less any amount realized from disposition is reflected in earnings. For financial statement purposes, property and equipment are recorded at cost and depreciated using the straight-line method over their estimated useful lives.
Advertising
The Company follows the policy of charging the costs of advertising to expense as incurred.
Income Taxes
Deferred income tax assets and liabilities are determined based on the estimated future tax effects of net operating losses, credit carryforwards, and temporary differences between the tax basis of assets and liabilities and their respective financial reporting amounts measured at the current enacted tax rates. The Company records an estimated valuation allowance on its deferred income tax assets if it is not more likely than not that these deferred income tax assets will be realized.
The Company recognizes a tax benefit from an uncertain tax position if it is more likely than not that the tax position will be sustained on examination by taxing authorities. Interest and penalties associated with such uncertain tax positions are classified as a component of income tax expense.
Stock-Based Compensation
Employee and non-employee share-based compensation is measured at the grant date, based on the fair value of the award, and is recognized as an expense over the requisite service period. Forfeitures are recognized as they occur.
The Company calculates basic net loss per common share by dividing the net loss available to common stockholders by the weighted-average number of shares of common stock outstanding during the period.
Diluted earnings per share is computed by giving effect to all potentially dilutive common stock equivalents in the period, including unvested stock options and warrants. As the Company has reported losses for all periods presented, all potentially dilutive securities have been excluded from the calculation of diluted net loss per common share as their effect would be antidilutive.
Recent Accounting Pronouncements
In January 2024, the Company adopted Accounting Standards Update (ASU) 2023-07, Improvements to Reportable Segment Disclosures (Topic 280). The new standard improves reportable segment disclosure requirements, primarily through enhanced disclosures about significant segment expenses that are regularly provided to the chief operating decision maker. ASU 2023-07 also clarifies that entities with a single reportable segment are subject to both new and existing reporting requirements under Topic 280. The adoption of this guidance did not result in a material effect on the Company’s financial statements. See the Segment Reporting section within Note 2.
| F-11 |
In December 2023, the FASB issued ASU No. 2023-09: Income Taxes (Topic 740): Improvements to Income Tax Disclosures that requires entities to disclose additional information about federal, state, and foreign income taxes primarily related to the income tax rate reconciliation and income taxes paid. The new standard also eliminates certain existing disclosure requirements related to uncertain tax positions and unrecognized deferred tax liabilities. The guidance is effective for the Company’s fiscal year ending December 31, 2025. The guidance does not affect recognition or measurement in the Company’s consolidated financial statements. For comparative purposes, the Company elected to present its 2024 income tax disclosures in this new manner, but the adoption of this guidance did not result in a material effect on the Company’s financial statements. See Note 9, Income Taxes.
In July 2025, the FASB issued ASU No. 2025-05 which amends Topic 326. Specifically, the ASU provides a practical expedient whereby an entity can assume that current conditions as of the balance sheet date will not change for the remaining life of the asset (e.g., the accounts receivable). This guidance is effective for the Company’s fiscal year ending December 31, 2026 and can be adopted early. The Company is in the process of evaluating the effects of this guidance on its consolidated financial statements.
In September 2025, the FASB issued ASU No. 2025-07 which, among other things, provides scope clarification for share-based noncash consideration from a customer in a revenue contract. Specifically, the ASU clarifies that share-based payments from customers in exchange for the transfer of goods or services should be accounted for as noncash consideration within the scope of ASC 606 as opposed to as a derivative pursuant to ASC 815 or as an equity security pursuant to ASC 321. This guidance is effective for the Company’s fiscal year ending December 31, 2027 and can be adopted early. The Company is in the process of evaluating the effects of this guidance on its consolidated financial statements.
The Company does not believe that any other recently issued effective pronouncements, or pronouncements issued but not yet effective, if adopted, would have a material effect on the accompanying financial statements.
3. Investments
The
Company’s sole investment is in the form of equity securities in a private entity. The Company entered into a strategic collaboration
agreement and received such equity securities as remuneration for services rendered. The investment is initially valued at approximately
$
4. Property and Equipment
Property
and equipment consisted of $
Depreciation
expense totaled $
5. Accrued Expenses
Accrued expenses consist of the following at December 31:
| 2025 | 2024 | |||||||
| Accrued payroll and related | $ | $ | ||||||
| Accrued legal fees | ||||||||
| Accrued licensing and royalty fees | ||||||||
| Accrued Board fees | ||||||||
| Accrued other expenses | ||||||||
| Total Accrued expenses | $ | $ | ||||||
| F-12 |
6. Revenue
During
2025, the Company had a collaboration agreement with a single customer, Eleison Pharmaceuticals, Inc. (“Eleison”), for
contract services. The collaboration agreement, which was entered into in February 2025 and designed to enhance clinical trial
efficacy, extract actionable insights from historical and ongoing data and improve strategic planning for Eleison’s oncology
pipeline, was deemed to have multiple deliverables with revenue to be recognized at the time each deliverable was completed. In
exchange for the services provided, the Company was entitled to consideration in the form of cash or equity securities of the
customer or any combination at the customer’s sole discretion. The Company received the initial payment in the second quarter
of 2025, representing 50% of the total consideration, in the form of equity securities of the customer valued at approximately
$
In June 2025, the Company entered into a strategic collaboration with Sygnature Discovery (“Sygnature”), a UK-based contract research organization specializing in drug discovery. Under this collaboration, Sygnature will introduce BullFrog Data Networks™, the Company’s proprietary AI-driven data insights platform powered by the bfLEAP™ engine, to Sygnature’s global biopharma client base. Any commercial terms for the marketing collaboration will be agreed by the parties in a subsequent agreement. The Company has not yet recognized any revenue under this collaboration.
The
Company currently has no other revenue agreements. Additionally, the Company has
7. Notes Payable
In
February 2024, the Company entered into an agreement to finance a portion of the premium for its directors and officers insurance policy
for the policy period of February 2024 through February 2025. The agreement provided for financing of $
In
February 2025, the Company again entered into an agreement to finance a portion of the premium for its directors and officers insurance
policy for the policy period of February 2025 through February 2026. The agreement provided for financing of $
8. Stockholders’ Equity
Preferred Stock
The
Company has shares of preferred stock authorized at a par value of $ with being designated as Series A Convertible
Preferred Stock. Of the authorized shares of Series A Convertible Preferred Stock, were issued and outstanding as of
December 31, 2025. Each share of Series A Convertible Preferred Stock is convertible at any time into
Common Stock
The Company has shares of common stock authorized at a par value of $.
In
February 2024, the Company received approximately $
| F-13 |
In
October 2024, the Company received approximately $
In
April 2025, the Company entered into the ATM Agreement with BTIG, LLC, pursuant to which the Company may offer and sell, from time to
time in its sole discretion, shares of common stock having an aggregate offering price of $
Through
December 31, 2025, the Company received approximately $
In
September 2025, the Company entered into a purchase agreement with Lincoln Park Capital Fund, LLC (“Lincoln Park”), pursuant
to which Lincoln Park committed to purchase up to $
Dilutive securities are excluded from the diluted earnings per share calculation because their effect is anti-dilutive. As of December 31, 2025, shares of preferred stock, warrants, options for shares of common stock, and unvested RSUs were excluded from the calculation of net loss per share. As of December 31, 2024, shares of preferred stock, warrants, and options for shares of common stock were excluded from the calculation of net loss per share. For the years ended December 31, 2025 and 2024, pre-funded warrants issued in 2020 as consideration for services of and , respectively, are included in the calculation of net loss per common share.
2022 Equity Incentive Plan
In November 2022, the Company’s Board of Directors adopted, and its shareholders approved, the 2022 Equity Incentive Plan (the “Plan”). The Plan provides for the granting of equity-based awards to employees, directors, and consultants. The Plan provides for equity-based awards including incentive stock options, non-qualified stock options, stock appreciation rights, performance share awards, cash awards and other equity-based awards. Awards are limited to a maximum term of years and any exercise prices shall not be less than 100% of the fair market value of one share of common stock on the grant date. The Plan authorized an initial maximum number of shares underlying awards of with an automatic annual increase to an amount equal to % of the total number of shares outstanding as of the end of the preceding fiscal year. In October 2025, the Company’s stockholders approved an amendment to the Plan to increase the number of shares available for issuance under the Plan by . As of December 31, 2025, there are awards authorized but unissued available under the Plan.
| F-14 |
Stock Options
Number of Shares | Weighted- Average Exercise Price | Weighted- Average Remaining Contractual Term (Years) | Aggregate Intrinsic Value | |||||||||||||
| Outstanding at December 31, 2023 | $ | | $ | |||||||||||||
| Granted | $ | |||||||||||||||
| Exercised | $ | |||||||||||||||
| Forfeited / canceled | ( | ) | $ | |||||||||||||
| Outstanding at December 31, 2024 | $ | $ | ||||||||||||||
| Granted | $ | |||||||||||||||
| Exercised | $ | |||||||||||||||
| Forfeited / canceled | ( | ) | $ | |||||||||||||
| Outstanding at December 31, 2025 | $ | $ | ||||||||||||||
| Vested at December 31, 2025 | $ | $ | ||||||||||||||
| 2025 | 2024 | |||||||
| Expected dividend yield | % | % | ||||||
| Expected volatility | % - % | % - % | ||||||
| Risk-free interest rate | % - % | % - % | ||||||
| Expected life (in years) | - | |||||||
| ● | Volatility – The trading volatility was determined by calculating the volatility of the Company’s peer group. | |
| ● | Expected life of options – The expected life of options granted to employees was determined using the simplified method. | |
| ● | Risk-free interest rate – This is the U.S. Treasury rate, having a term comparable to the expected life of the stock option. | |
| ● | Dividend yield – The Company does not expect to pay a dividend in the foreseeable future. |
The weighted-average grant-date fair value of options granted during the years ended December 31, 2025 and 2024 was $ and $, respectively. The total grant-date fair value of options vested during the years ended December 31, 2025 and 2024 was approximately $ and $, respectively.
During the year ended December 31, 2024, certain stock option awards granted to related parties were modified as follows:
| ● | In July 2024, the Company modified vested stock option awards granted to the former Chief Marketing Officer by extending his period to exercise these awards from 6 months to 12 months following termination. This modification resulted in additional stock-based compensation expense of $. |
| ● | In December 2024, the Company modified stock option awards granted to the former Chief Financial Officer by (i) accelerating the vesting of unvested options, and (ii) extending the period to exercise all now-vested awards from 6 months to 12 months following his passing. These modifications resulted in additional stock-based compensation expense of $. |
No options were exercised in any of the periods presented.
During the years ended December 31, 2025 and 2024, the Company recognized approximately $ and $, respectively, of compensation expense related to stock options.
| F-15 |
As of December 31, 2025, the total unrecognized compensation expense related to unvested stock options was approximately $, which the Company expects to recognize over a weighted-average period of approximately year.
Restricted Stock Units
During the year ended December 31, 2025, the Company granted restricted stock units (“RSUs”) with an average grant date fair value of $. The RSUs vest over a two-year period. During the year ended December 31, 2025, the Company recognized $ of compensation expense related to RSUs.
As of December 31, 2025, the total unrecognized compensation expense related to unvested RSUs was approximately $, which the Company expects to recognize over a weighted-average period of approximately years.
Stock Grants
During the year ended December 31, 2025, the Company granted shares of common stock with an average grant date fair value of $, all of which were fully vested at the grant date and all of which were issued prior to December 31, 2025. During the year ended December 31, 2025, the Company recognized $ of compensation expense related to the stock grants.
Warrants
The following table provides details of the Company’s outstanding warrants including those issued as consideration for services and those issued in conjunction with transactions as of December 31, 2025:
| Exercise Price | Expiration | Number of Warrants | ||||
| $ | ||||||
| $ | ||||||
| $ | ||||||
| $ | ||||||
| $ | ||||||
| $ | ||||||
Warrants Issued in Conjunction with Transactions
During the year ended December 31, 2024, the Company issued the following warrants as part of two equity offerings:
| ● | In
February 2024, |
| ● | In
February 2024, pre-funded warrants with an exercise price of $ |
| ● | In
February 2024, |
| ● | In
October 2024, |
| F-16 |
| ● | In
October 2024, pre-funded warrants with an exercise price of $ |
| ● | In
October 2024, |
Warrants Issued as Consideration for Services
The following table summarizes the activity for warrants issued as consideration for services for the years ended December 31, 2025 and 2024:
Number of Warrants | Weighted- Average Exercise Price | Weighted- Average Remaining Contractual Term (Years) | Aggregate Intrinsic Value | |||||||||||||
| Outstanding at December 31, 2023 | $ | | $ | |||||||||||||
| Granted | $ | |||||||||||||||
| Exercised | ( | ) | $ | |||||||||||||
| Forfeited / canceled | $ | |||||||||||||||
| Outstanding at December 31, 2024 | $ | $ | ||||||||||||||
| Granted | $ | |||||||||||||||
| Exercised | $ | |||||||||||||||
| Forfeited / canceled | $ | |||||||||||||||
| Outstanding at December 31, 2025 | $ | $ | ||||||||||||||
| Vested at December 31, 2025 | $ | $ | ||||||||||||||
During the years ended December 31, 2025 and 2024, the Company recognized $ and $, respectively, of compensation expense related to certain warrants.
As of December 31, 2025, there was $ of unrecognized compensation expense related to certain warrants, which the Company expects to recognize over a weighted-average period of approximately years.
The total grant-date fair value of warrants vested during the year ended December 31, 2025 was approximately $.
| F-17 |
9. Income Taxes
Deferred income taxes reflect the net tax effects of net operating losses, credit carryforwards, and temporary differences between the carrying amounts of assets and liabilities for financial reporting purposes and the amounts used for income tax purposes. Significant components of the Company’s deferred tax assets for federal and state income taxes are as follows:
| December 31, | ||||||||
| 2025 | 2024 | |||||||
| Deferred tax assets: | ||||||||
| Net operating losses | $ | $ | ||||||
| Capitalized research and development | ||||||||
| Stock-based compensation | ||||||||
| Intangibles | ||||||||
| Other | ||||||||
| Total deferred tax assets | ||||||||
| Valuation allowance | ( | ) | ( | ) | ||||
| Net deferred tax asset | ||||||||
| Deferred tax liabilities: | ||||||||
| Property and equipment | ( | ) | ( | ) | ||||
| Total deferred tax liabilities | ( | ) | ( | ) | ||||
| Net deferred tax asset / (liability) | $ | $ | ||||||
Realization
of the Company’s deferred tax assets is dependent upon future earnings, if any, the timing, and amount of which are uncertain.
Because of the Company’s lack of U.S. earnings history, the net U.S. deferred tax assets have been fully offset by a valuation
allowance. The valuation allowance increased by $
As
of December 31, 2025, the Company has available for federal income tax purposes a gross net operating loss carryforward of approximately
$
The Company has provided a valuation allowance against the full amount of the net operating loss benefit and its other net deferred tax assets since, in the opinion of management, based upon the earnings history of the Company, it is more likely than not that the future tax benefits of the Company’s net deferred tax assets will not be realized. All or a portion of the remaining valuation allowance may be reduced in future years based on an assessment of earnings sufficient to fully utilize these potential tax benefits.
As required under ASU 2023-09, the Company has included only the portion of the valuation allowance related to federal deferred tax assets in the “change in valuation allowance” line of the rate reconciliation. The following table presents a reconciliation of the total change in the valuation allowance:
| December 31, | December 31, | |||||||
| 2025 | 2024 | |||||||
| Beginning balance | $ | $ | ||||||
| Change charged to income tax expense | ||||||||
| Ending balance | $ | $ | ||||||
The Company has incurred net operating losses since inception and it did not have unrecognized tax benefits as of December 31, 2025 and 2024, and does not anticipate this to change significantly over the next 12 months. The Company will recognize interest and penalties accrued on any unrecognized tax benefits as a component of income tax expense.
The Company files income tax returns in the U.S. and certain state jurisdictions. The Company is not currently under examination in these jurisdictions for any tax year. The Company’s tax years beginning with 2022 are open tax years. Because of net operating losses and research credit carryovers, substantially all the Company’s tax years remain open to examination.
| F-18 |
Income tax provision (benefit) related to continuing operations differs from the amounts computed by applying the statutory income tax rate of 21% to pretax loss as follows:
| December 31, 2025 | December 31, 2024 | |||||||||||||||
| Amount | Rate | Amount | Rate | |||||||||||||
| U.S. Federal statutory tax rate | $ | ( | ) | % | $ | ( | ) | % | ||||||||
| Change in valuation allowance | ( | ) | ( | ) | ||||||||||||
| Stock-based compensation | ) | ) | ||||||||||||||
| Other | ( | ) | ||||||||||||||
| $ | % | $ | % | |||||||||||||
10. Material Agreements
JHU-APL Technology License
In
February 2018, the Company entered into an exclusive, world-wide, royalty-bearing license with JHU-APL (the “2018 License Agreement”).
The license covers three (3) issued patents, one (1) new provisional patent application, non-patent rights to proprietary libraries of
algorithms and other trade secrets, as well as modifications and improvements. In October 2021, the Company executed an amendment to
the original license for improvements and new advanced analytics capabilities. In consideration of the rights granted to the Company
under the 2018 License Agreement, JHU-APL received a warrant equal to five percent (
In
July 2022, the Company entered into an exclusive, world-wide, royalty-bearing license from JHU-APL for the additional technology developed
to enhance the bfLEAP™ platform (the “2022 License Agreement”). The new license provides additional intellectual property
rights including patents, copyrights, and know-how to be utilized under the Company’s bfLEAP™ analytical AI/ML platform.
This 2022 License Agreement supersedes the previous 2018 License Agreement. In consideration for entering into the new license, the Company
issued shares of common stock to JHU-APL. Under the terms of the 2022 License Agreement, JHU-APL will be entitled to eight percent
(
In
May 2023, the Company and JHU-APL entered into Amendment Number 1 of the 2022 License Agreement whereby the Company gained access to
certain improvements including additional patents and know-how in exchange for a series of payments totaling $
As
of December 31, 2025, the Company has accrued the entire $
George Washington University - Beta2-spectrin siRNA License
In January 2022, the Company entered into an exclusive, world-wide, royalty-bearing license from George Washington University (“GWU”) for rights to use siRNA targeting Beta2-spectrin in the treatment of human diseases, including hepatocellular carcinoma. The license covers methods claimed in three U.S. and worldwide patent applications, and also includes use of this approach for treatment of obesity, non-alcoholic fatty liver disease, and non-alcoholic steatohepatitis.
| F-19 |
In
consideration of the rights granted to the Company under the license agreement, the Company paid GWU a $
Johns Hopkins University – Mebendazole License
In February 2022, the Company entered into an exclusive, world-wide, royalty-bearing license from Johns Hopkins University (“JHU”) for the use of an improved formulation of Mebendazole for the treatment of any human cancer or neoplastic disease. This formulation shows potent activity in animal models with different types of cancer and has been evaluated in a Phase I clinical trial in patients with high-grade glioma (NCT01729260). The trial, an open-label dose-escalation study, assessed the safety and efficacy of the improved formulation with adjuvant temozolomide in 24 patients with newly diagnosed gliomas. Investigators observed no dose-limiting toxicity in patients receiving all but the highest tested dose (200mg/kg/day). Four of the 15 patients receiving the maximum tested dose of 200mg/kg/day experienced dose-limiting toxicity, all of which were reversed by decreasing or eliminating the dose given. There were no serious adverse events attributed to Mebendazole at any dose during the trial. 41.7% of patients who received Mebendazole were alive at two years after enrollment, and 25% were alive at four years (Gallia et al., 2021).
The
license covers six (6) issued patents and one (1) pending application. In consideration of the rights granted to the Company under the
license agreement, JHU received a staggered upfront license fee of $
Johns Hopkins University – Prodrug License
In
October 2022, the Company entered into an exclusive, world-wide, royalty-bearing license from JHU and the Institute of Organic Chemistry
and Biochemistry (“IOCB”) of the Czech Academy of Sciences for rights to commercialize N-substituted prodrugs of Mebendazole
that demonstrate improved solubility and bioavailability. The license covers prodrug compositions and use for treating disease as claimed
in multiple U.S. and worldwide patent applications. In consideration for the rights granted to the Company under the license agreement,
JHU and IOCB received a staggered upfront license fee of $
| F-20 |
Lieber Institute for Brain Development Partnership
In September 2023, the Company entered into a Data Use and Technology Partnership Agreement (the “Data Use Agreement”) and a related Memorandum of Understanding (“MOU”) with the Lieber Institute for Brain Development (“LIBD”), a nonprofit medical research organization focused on mental health disorders. The partnership is intended to combine LIBD’s proprietary brain-related datasets with the Company’s AI/ML capabilities to support drug discovery and development activities.
Under the Data Use Agreement, LIBD granted the Company a limited, royalty-free, non-transferable license to access and use certain curated LIBD datasets solely for the application of AI/ML to drug development, excluding diagnostic uses. The license was exclusive for an initial one-year term beginning upon receipt of the first significant tranche of data and was subsequently extended. The Company is responsible for all costs associated with the development plan and is required to provide LIBD with the resulting deliverables upon completion of the exclusivity period.
Pursuant to the MOU, the Company entered into a Commercial Agreement with LIBD governing the potential commercialization of products or services derived from LIBD data. Under the proposed structure, LIBD would receive royalties on net sales and a percentage of sublicense revenue, with rates varying based on the source of commercialization. The Company has also agreed to provide LIBD with any such products or services free of charge for LIBD’s internal research use.
11. Commitments and Contingencies
While not assured, management does not believe, based upon information available at this time, that a loss contingency will have a material adverse effect on the Company’s financial position, results of operations or cash flows. Additionally, the Company does not have any material commitments.
12. Subsequent Events
The Company evaluates subsequent events and transactions that occur after the balance sheet date up to the date that the consolidated financial statements are issued.
Subsequent
to the year ended December 31, 2025, the Company continued to raise capital through sales of shares of its common stock under its
ATM Agreement. Subsequent to December 31, 2025, the Company received approximately $
Subsequent
to the year ended December 31, 2025, the Company raised capital through its purchase agreement with Lincoln Park. In January 2026, the
Company received proceeds of approximately $
| F-21 |